Manisit Das
Breast cancer may sound like a single disease, but it is not. There are many subtypes of the disease, which guide the course of disease progression and treatment strategy. One of these subtypes, referred to as triple-negative breast cancer (TNBC) is particularly difficult to treat. Recently, researchers at Lund University, Sweden identified a cellular growth factor that may be linked to this aggressive TNBC subtype. Blocking the growth factor in experimental cancer models transformed the tumor into a subtype which is known to have a better clinical outcome. Down the road, this can be a unique therapeutic strategy against TNBC.
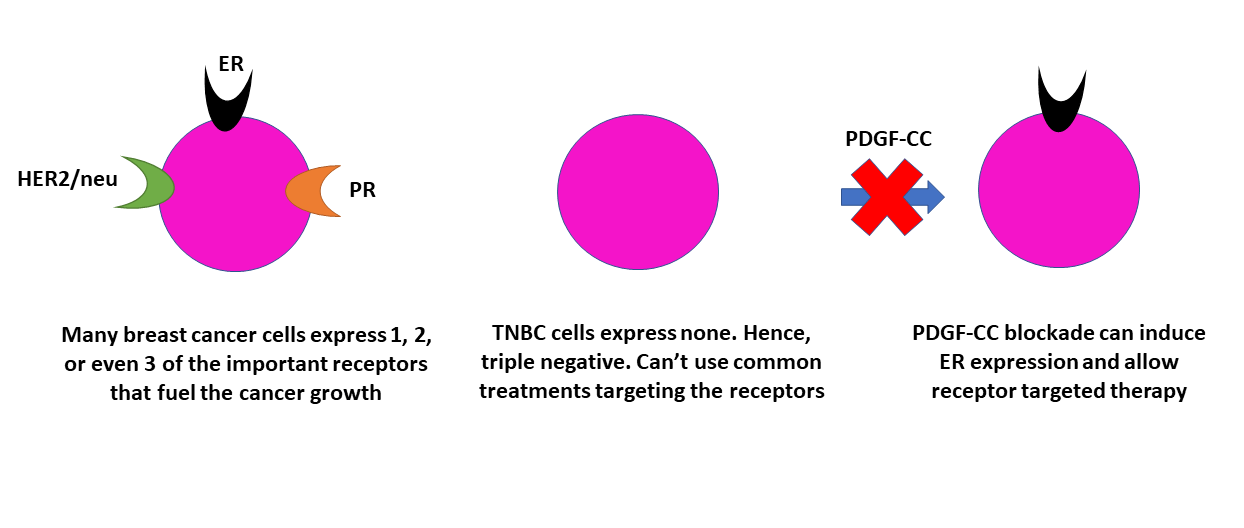
 A critical step after a diagnosis of breast cancer is to determine the molecular subtype of the tumor. Classification of the tumors is guided by the expression of hormone epidermal growth factor receptor 2 (HER2), estrogen receptors (ER), and progesterone receptors (PR). Testing positive for one of these receptors may make a patient eligible for hormone therapy* or drugs that target a specific receptor, such as HER2. TNBC lacks these receptors, making hormone or receptor-targeted therapies ineffective against this type of cancer. Chemotherapy and radiation are the most common treatments for TNBC patients. In a study recently published in Nature Medicine, senior author Kristian Pietras and his team identified a strategy to change the molecular subtype of TNBC to ER-positive tumors, rendering the cancer sensitive to Tamoxifen, a drug commonly used to treat estrogen receptor-positive breast cancer.
A critical step after a diagnosis of breast cancer is to determine the molecular subtype of the tumor. Classification of the tumors is guided by the expression of hormone epidermal growth factor receptor 2 (HER2), estrogen receptors (ER), and progesterone receptors (PR). Testing positive for one of these receptors may make a patient eligible for hormone therapy* or drugs that target a specific receptor, such as HER2. TNBC lacks these receptors, making hormone or receptor-targeted therapies ineffective against this type of cancer. Chemotherapy and radiation are the most common treatments for TNBC patients. In a study recently published in Nature Medicine, senior author Kristian Pietras and his team identified a strategy to change the molecular subtype of TNBC to ER-positive tumors, rendering the cancer sensitive to Tamoxifen, a drug commonly used to treat estrogen receptor-positive breast cancer.
Tumor cells interact strongly with the surrounding environment, referred to as the tumor microenvironment, by transmitting and receiving survival and growth signals. In their work, Dr. Pietras and his team identified a protein of the platelet-derived growth factor family (PDGF-CC), which facilitate communication between cancer and surrounding connective tissue cells like fibroblasts.
Analyzing genetic information from tissue samples of about 900 breast cancer patients, they discovered a strong connection between PDGF-CC expression and poor clinical outcome. To confirm if PDGF-CC expression is important in the progression of breast cancer, the researchers developed a genetically engineered mouse model where they artificially disrupted the expression of PDGF-CC and looked at tumor growth. This way of knocking out a gene is a strategy commonly used by biologists to understand the function of a gene. They observed a poorer tumor growth when mice lacked the PDGF-CC gene. To ensure that this is not due to other developmental defects in the engineered mice, the researchers transplanted fragments of the tumor from the PDGF-CC defective mice to non-engineered mice. About half of the transplants didn’t give rise to tumors. In comparison, transplants from mice which didn’t have the PDGF-CC gene knocked out formed tumors with every attempt.
The researchers next looked into how PDGF-CC expression affects the expression of different genes in the breast tumors. From their experiments, they found that PDGF-CC expression plays a role in influencing the molecular subtype of the breast cancer. They uncovered a positive correlation between PDGF-CC expression and markers of TNBC. This led them to their subsequent experiment, where they tested if blockade of PDGF-CC can switch the subtype of breast cancer.
An antibody was used to block PDGF-CC. Antibodies are large proteins which can be engineered to bind and inhibit other target proteins of interest. Blocking PDGF-CC led to increase in the expression of ER, which indicated a switch in cancer subtype. The inhibition of PDGF-CC by gene knockout or antibody also made the breast tumors sensitive to tamoxifen therapy. The therapeutic benefit was lost when the PDGF-CC gene was artificially introduced into the mice.
It was originally believed that the various subtypes of breast cancer originate from different cells in the mammary gland. However, this study shows that cells in the tumor microenvironment can also directly influence the subtype of cancer, and is tunable by pharmaceutical intervention. This is indeed an exciting development. Nevertheless, there is a considerable journey ahead before this becomes a standard clinical practice. As we are gaining more insights into molecular characteristics of cancer, it is also increasingly evident that there is no such thing as a pure cancer subtype, thanks to intratumor heterogeneity. In other words, there can be considerable variations of cancer cells within a single tumor. Under the circumstances, subtype switching may sensitize one group of cells within the tumor, while not affecting another group of cells. We do not yet know if the cells under PDGF blockade would eventually become resistant by exploiting a compensatory signaling. Thus, there are a lot of questions still to be answered.
TNBC is a particularly aggressive form of cancer more likely to affect young people, African-Americans, Hispanics, and people with inherited mutations that make them susceptible to breast cancer. We sincerely hope that the findings of this study will stimulate a new direction in designing a clinical strategy for patients affected by this aggressive form of breast cancer.
*Author’s note: Hormone therapies for breast cancer slow down the growth of the tumor by cutting off the hormonal supply of the cancer cells. The hormones fuel the growth of hormonal receptor-positive cancers. This strategy is not same, rather opposite of hormone replacement therapy (HRT) used by postmenopausal women to ease symptoms of menopause. In fact, HRT may increase the likelihood of breast cancer.
Work discussed:
Roswall, P., Bocci, M., Bartoschek, M., Li, H., Kristiansen, G., Jansson, S., . . . Pietras, K. (2018). Microenvironmental control of breast cancer subtype elicited through paracrine platelet-derived growth factor-CC signaling. Nature Medicine. doi: 10.1038/nm.4494