Reading time: 4 minutes
Aishwarya Subramanian
At Oncobites we’ve covered the exciting emerging field of immunotherapy to treat cancer, some of which include PROTACs and BiTE molecules . Recently, significant advances have been made when trying to come up with cutting edge ideas for cancer treatment. With the recent changes in the landscape of cancer immunotherapy there has been great progress that helps to contain or treat cancer. We have seen development of therapeutics that regulate critical checkpoints involved in cell division and cell death; engineering of T cells that target cancer cells effectively and help to control the cancer spread.
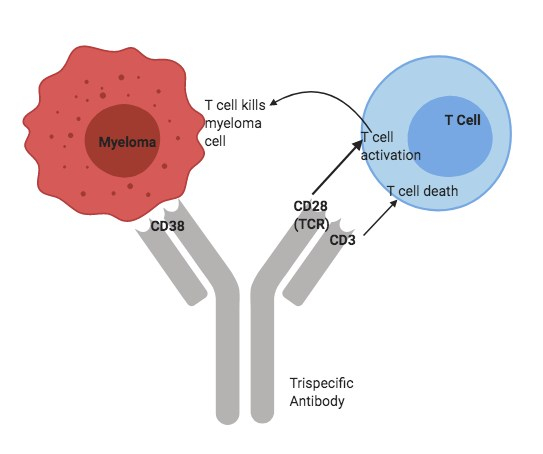
With the constant development of new treatment strategies against cancer there still remains a never ending challenge to treat certain types of malignant cancer such as Multiple Myeloma. Recently, it has been shown that the trispecific antibody platform has shown to be a very successful approach to a few critical targets. The trispecific antibody is designed in such a way that one antibody (with the characteristics of three separate antibodies) is sufficient enough to engage three different targets and mount a successful and effective immune response.
Wu et al. recently came up with the strategy to try and use the trispecific antibody platform as an approach to target cancer that will lead to increased T cell response to eliminate cancer cells and in turn prevent tumor growth. They engineered a trispecific antibody that has three targets – a cancer cell, a receptor that activates T cells, and a protein that promotes long lasting T cell activity against the cancer cell (1). This method of engineering a trispecific antibody helps to boost T cell function against cancer cells and achieve efficient response by stimulating the receptors to positively boost the T cell response.
Previous research has shown that two signals are required to activate T cells(3). Activation that occurs through CD3 (co-receptor) complex leads to increased cytokine secretion and activation through CD28 ensures that apoptotic (programmed cell death) function is inhibited. In the absence of the second signal, CD28 can cause activation of programmed cell death. Therefore, using these two characteristic functions the trispecific antibody was engineered.
By engineering this trispecific antibody, Wu et al. have used it to target cancer cells against multiple myeloma. When T cells recognise the presence of the antigen through the T cell receptor (CD28), the T cell receptor acts as an additional co-stimulatory signal that is essential to achieve the sustained prolonged T cell proliferation required for an effective immune response.
The overall idea of engineering an antibody to bind three targets is to add a co-stimulatory receptor that will help for a long-lasting T cell activity in presence of cancer cells.
It has also been shown that CD28 is present as an antigen marker in certain cancer cells such as leukemia and multiple myeloma. Using this function, it will help to effectively increase the binding of T cells to CD28 that’s present in the cancer cell. This especially helps in cases where the surface antigen on the cancer cell is either masked or present at very low quantities due to previous cancer treatments(2).
Edited by Tamara Vital
Works Discussed:
- Wu, L., Seung, E., Xu, L. et al. Trispecific antibodies enhance the therapeutic efficacy of tumor-directed T cells through T cell receptor co-stimulation. Nat Cancer (2019) doi:10.1038/s43018-019-0004-z
- Trispecific antibodies offer a third way forward for anticancer immunotherapy- Alfred L. Garfall & Carl H. June https://doi.org/10.1038/d41586-019-03495-3
- Esensten, J. H., Helou, Y. A., Chopra, G., Weiss, A., & Bluestone, J. A. (2016). CD28 Costimulation: From Mechanism to Therapy. Immunity, 44(5), 973-988. doi: 10.1016/j.immuni.2016.04.020
Featured image created using Biorender

Leave a comment