Reading time: 5 minutes
Tamara Vital
How do you stop an accident from happening? You could try to prevent the conditions that contribute to accidents by establishing rules that encourage and reward caution and safe behavior. On the road, licensing laws, traffic lights, and speed limits set the rules of the road, and those who break the rules must pay the consequences in tickets and fines. Together, these approaches help prevent accidents and allow drivers to arrive at their destinations safely. But if an accident is imminent, what can you do? In your car, you might slam on the breaks to decrease the impact of the crash and hope to minimize the damage.
In many ways, cancer treatment is similar. Ideally, we would like to be able to prevent cancer from happening in the first place by preventing the conditions that contribute to cancer. We hope to do this by engaging in safe behaviors like wearing sunscreen, avoiding smoking, and eating a healthy diet. However, despite our best efforts, not all tumors can be prevented. For the cancers that aren’t prevented, we want to be able to slam the breaks and not only stop the tumor, but destroy it entirely. At the molecular level, hitting the breaks has traditionally meant chemotherapy which stops the cellular processes that tumors need to sustain themselves and grow. Unfortunately, because these processes are also important for many normal cells targeting them leads to the collateral damage of chemotherapy side effects. More recently, by better understanding the molecular drivers of certain tumors, targeted therapies have been developed. These targeted therapies specifically target the proteins responsible for promoting cancer and inhibit the functions of the protein that contributes to the tumor.
Targeted therapies are the goal in cancer therapeutics in part because they are a major improvement for patients– by targeting a single mutated protein, targeted therapies may decrease side effects. Targeted therapies represent a rational strategy for treating cancer: stop the driver, stop the tumor. If targeted inhibitors are so great, why are we still using chemotherapy at all? Simply put, targeted therapies require good targets. Even for the tumors with well understood drivers, the functional part of those driver proteins might be difficult to target with drugs. These barriers to druggability are a major obstacle to developing targeted therapies to known or expected targets in cancer. So if you can’t target and inhibit a mutated driver protein, what can you do? What if instead of stopping the protein you could get rid of the protein all together?
Recently, strategies to overcome the difficulties in targeting the functional surfaces of mutated proteins have instead exploited the cell’s protein disposal systems to target and destroy tumor drivers. The strategy, proteolysis targeting chimeras (PROTACs) is elegant, but has multiple moving parts: the target protein, a bifunctional or chimeric chemical, and the ubiquitin-proteosome system. The bifunctional chemical is designed to have two functions: targeting both the mutated tumor driver and an E3 ligase, a member of the ubiquitin-proteosome system, the cell’s protein destruction system. E3 ligases modify other proteins (substrates) by adding ubiquitin, another small protein, to their substrates. The ubiquitin-tagged substrate can then be recognized by the other members of the ubiquitin-proteosome system which target the substrate to the proteosome where it is destroyed. By making the target protein a new substrate of an E3 ligase, PROTACs work by co-opting the process by which cells destroy proteins to destroy the target protein instead of inhibiting it.
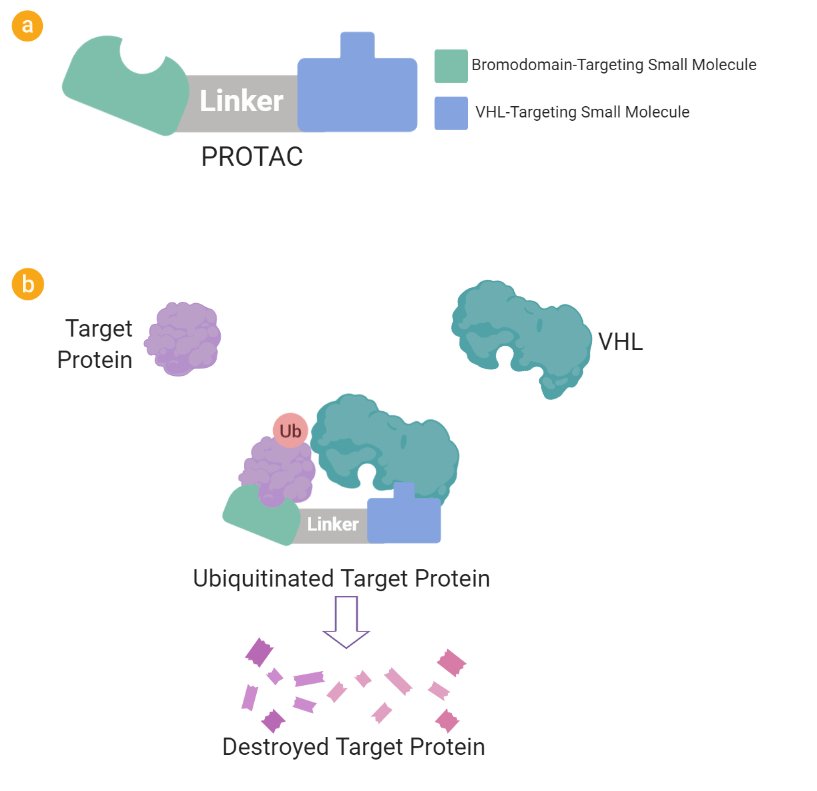
b. Destruction of a Target protein using a PROTAC. The PROTAC interacts with both the Target Protein (purple) and the E3 ligase (VHL, dark green). The interaction of the PROTAC with the Target and VHL leads to ubiquitination of the Target. The ubiquitinated Target is then degraded by the cell’s protein destruction machinery
In a recent paper in Nature Chemical Biology, Farnaby, Koegl, and colleagues generate a PROTAC targeting members of the BAF complex, an important protein complex whose members are commonly mutated in cancer. To generate this PROTAC, the authors first identified a small molecule to target a specific protein domain, a bromodomain, that is shared among important BAF complex members. Then they identified a small molecule to recruit VHL, an E3 ligase. After having identified small molecules to target both the protein to be degraded and the E3 ligase to tag the protein for destruction, the authors then sought to find a linker to bring together these two small molecules while preserving their functions and promoting physical interactions to facilitate ubiquitination of the target protein. The authors determined how the small molecules bound to the target proteins which allowed them to use a rational design strategy to identify a small molecule that binds to both the target protein and VHL to degrade the target proteins that contain the specifically targeted protein domain: SMARCA2, SMARCA4, and PBRM1. Because these proteins are commonly mutated in cancer, the authors examined whether the PROTAC could inhibit the growth of cancer cells. The authors found that while the original bromodomain-targeting small molecule did not have an effect on cancer cells, the PROTAC not only slowed cancer cell growth, but also induced cancer cell death. These experiments indicate that previously identified small molecules with known targets but limited biological potency can be used to create PROTACs with significant and specific effects against cancer cells.
PROTACs are still in the early stages of development, but they are already proving to be an attractive strategy for targeting mutated proteins in cancer. To make the leap from interesting tool to cancer drug, more work will need to be done to better understand how to develop PROTACs which effectively target and degrade problematic proteins. Critically, this work will require understanding what surfaces of the target protein and the E3 ligase need to be brought close together to ensure ubiquitination and ultimate destruction of the target and further work will be required to identify E3 ligases that are best suited for each target-small molecule pair. Still, PROTACs represent the latest effort to find chemicals that will hit the brakes on cancer.
Edited by Nisitha Sengottuvel
Work Discussed:
Farnaby, W., Koegl, M., Roy, M.J. et al. BAF complex vulnerabilities in cancer demonstrated via structure-based PROTAC design. Nat Chem Biol15, 672–680 (2019) doi:10.1038/s41589-019-0294-6
Header Image Credits: Eric Parker

