Reading time: 4 minutes
Sara Musetti
If you’ve heard of collagen, it’s likely been in articles raving about its benefits. Maybe it was in the recent collagen broth craze, when everyone from Halle Berry to Dr. Oz claimed that the collagen in bone broth could prevent aging, improve your hair, cure arthritis, and so on. While these particular claims are complete nonsense, the importance of collagen to your health is indisputable. Collagen is a long, ropy protein that makes up your hair, skin, cartilage, and many other tissues; it is absolutely vital to your life, and is the most abundant protein in your body. It also helps your skin keep its shape and resist wrinkles, so it’s often hyped up in skin care. But, like so many things, cancer manages to take this lovely feature and turn it into a nightmare.
Collagen is used to support and protect, so naturally tumors twist it to their advantage. Tumors are often full of fibroblasts, the major cell type responsible for producing collagen. These cells pump out huge amounts of collagen, swadling little pockets of tumor cells, called tumor nests, in blankets of collagen that keep damaging agents away. These collagen-rich regions form a physical barrier around tumor cells that keep chemotherapeutics, immune cells, antibodies, and other therapies from reaching the cells to kill them. The particular shape and character of collagen in a tumor has even been linked to how easily the tumor grows and spreads. So usually, when cancer researchers are discussing cancer, it’s in a frustrated tone: How do we get around collagen to kill the tumor cells?
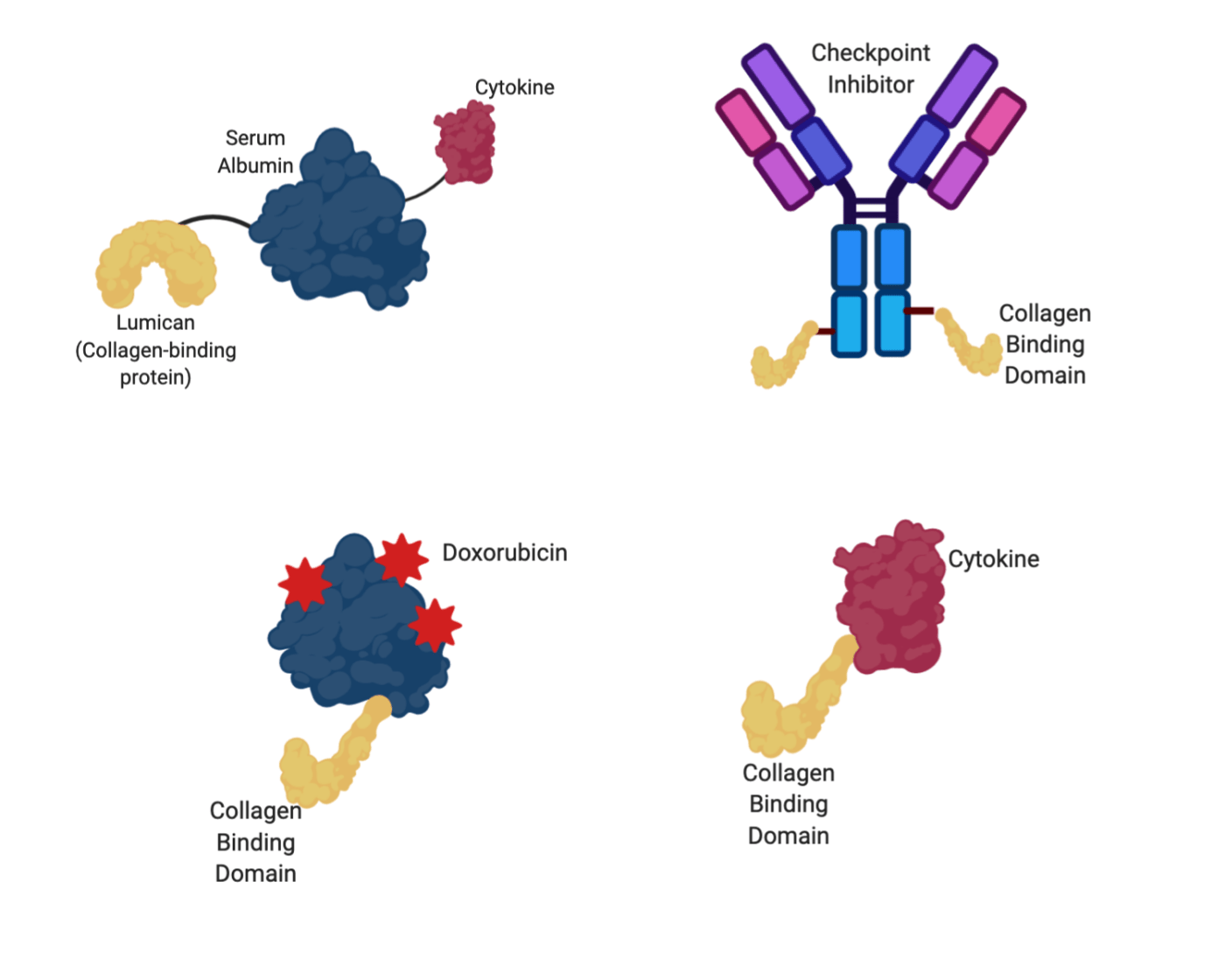
A recent trend in cancer drug delivery has been to stop treating collagen like the problem and start treating it like part of the solution. If we know tumors are collagen rich, and that most tumors have leaky blood vessels helping drugs and other particles to exit circulation draining into tumors, why not use those attributes to kill tumors?? Researchers in two separate groups, one at MIT’s Koch Institute and one at the University of Chicago’s Pritzker School of Molecular Engineering, have found ways to tether therapies to collagen-binding proteins (yellow, Figure 3) and increase the tumor payload. One method from MIT uses a common blood protein, known as serum albumin (blue, Figure 3), to carry the chemotherapy drug doxorubicin (bright red, Figure 3) to the tumor. Because tumors tend to be more acidic than other tissues, the doxorubicin was bound to its carrier with an acid-cleavable bond, so that when the doxorubicin-loaded protein is attracted to a tumor and binds to collagen, doxorubicin is released. A second method, from U Chicago, attaches an immune-stimulating cytokine (dark red, Figure 3), a signaling molecule, to a collagen-binding protein. By increasing the amount of anti-tumor immune cells within the tumor, these collagen-binding proteins were able to boost the immune system with the tumor enough that CAR-T therapies could be used to fight the tumor, something that has been notoriously difficult in solid tumors. Additionally, collagen binding domains can be used to help retain antibodies, like checkpoint inhibitors that help the immune system fight cancer, within the tumor for longer periods and at higher concentrations, making them more effective.
Compared with untethered doxorubicin, checkpoint inhibitors, or cytokines, these new collagen-binding therapies are far more effective at curing a variety of tumors in animal models, because they are far less toxic, and tend to localize within the tumor rather than in other tissues. This lessens the extensive liver and kidney damage. Targeting drugs to tumor proteins is not new, but this new trend does seem to be significant, in that many tumors express high amounts of collagen, so this particular target can be used for many hard-to-treat tumors.
So far, these studies have only been done in mice, so a great deal more work must be done before they can be available to patients, if they are successful in humans. However, while these particular therapies are not yet ready for the clinic, the trend of using collagen as a target may help boost the potency of many therapies that are already on the market. As always in medicine, the first step is the hardest, and knowing that the curse of collagen can be turned around is a beacon of hope for many researchers.
Edited by Payal Yokota
Works Cited
Momin, N., Mehta, N. K., Bennett, N. R., Ma, L., Palmeri, J. R., Chinn, M. M., … & Wittrup, K. D. (2019). Anchoring of intratumorally administered cytokines to collagen safely potentiates systemic cancer immunotherapy. Science Translational Medicine, 11(498).
Sasaki, K., Ishihara, J., Ishihara, A., Miura, R., Mansurov, A., Fukunaga, K., & Hubbell, J. A. (2019). Engineered collagen-binding serum albumin as a drug conjugate carrier for cancer therapy. Science advances, 5(8).
Ishihara, J., Ishihara, A., Sasaki, K., Lee, S. S. Y., Williford, J. M., Yasui, M., … & Raczy, M. M. (2019). Targeted antibody and cytokine cancer immunotherapies through collagen affinity. Science translational medicine, 11(487).
Image Credits
Figure 1: Structure of collagen strands. © Eric Hahn. https://commons.wikimedia.org/wiki/File:Collagen_(triple_helix_protein_with_schematic_ribbons).jpg
Figure 2: Collagen (light pink) surrounds tumor nests (purple/dark pink). © Nephron via Wikimedia. https://commons.wikimedia.org/wiki/File:Desmoplastic_small_round_cell_tumour_-_intermed_mag.jpg (header image)
Figure 3: Various collage-binding therapies used in these studies to treat cancer. Albumin, the carrier protein, is shown in blue. Therapeutic agents are shown in red (doxorubicin=bright red, cytokines=dark red), while collagen-binding domains are shown in yellow. Image created in BioRender by the author.

Does this mean collagen powder can cause tumors in women?
LikeLike
While I do not know of any studies on this particular question, it’s extremely unlikely that collagen powder would be able to induce tumors. For starters, it is far too large a molecule to pass efficiently from the GI tract to other organs when ingested. Additionally, much of the danger of collagen in tumors comes from its location; in tumors, it forms nests that surround the tumor and prevent drugs from effectively killing the cancer cells. Powdered collagen would not form a similar structure and would therefore not pose the same risks.
Best,
Sara
LikeLike
Hi, I am taking Anastrozole after having had breast cancer. I have recently been taking marine collagen capsules for my joint health. Is it ok to take Anastrazole and collagen together? Thank you 😊
LikeLike
Hi Linda, thank you for reaching out. We are not licensed clinicians, and we do not have the authority to make any treatment recommendations. Please consult with your physician about your treatment. We, however, appreciate you reading our blog and wish you our best. Take care! -Team OncoBites
LikeLike
HelloI I have stage 3 Papillary cancer. A friend gifted me a Neocell collagen. Should I take it?
LikeLike
Hi, sorry for the delayed response. We suggest asking your healthcare provider about this. We cannot provide medical advice in this forum. Thank you!
LikeLike