Reading time: 4 minutes
Zoey Tang
In the world of anticancer therapy, there are a lot of drugs that belong to a class of proteins called monoclonal antibodies (mAbs) These Y-shaped proteins can tightly hug other proteins- including disease targets on tumor cells- with both of their arms, making the tumor cells die. Moreover, unlike some not-so-specific anticancer agents, mAbs only hug the targets they like, causing less uncomfortable side effects in cancer patients. Because of the high treatment efficacy and minimal toxicity effects, mAbs are regarded as “magic bullets.” Over 30 mAbs have been approved by the US or EU in anticancer treatment. It’s safe to say that mAb buzzed in the anticancer treatment field.
However, not all solid tumor patients benefit from mAb therapies. For example, cetuximab (an mAb against a protein called epidermal growth factor receptor (EGFR)- which is found on high levels in cancer cells allowing them to grow and divide) monotherapy only has a 13% overall response rate in patients with squamous cell carcinoma of the head and neck- a type of skin cancer. One of the reasons for the low response rate is that we do not fully understand how mAbs work in solid tumors. Like other agents, mAbs can only elicit pharmacological effects when they are engaged to their targets. Therefore elucidating the hug between mAbs and their targets (or “mAb-target engagement”) is a critical step to understanding the mAb tumor-killing effect.
Attempts have been made to study mAb-target engagement in living animals. For example, mAbs are labeled with fluorophores – light tracers– before injecting into animals or human patients. It’s like assigning each of the solider a GPS tracker to see where they are at the battlefield. The distribution of mAbs in living animals/ patients is then visible by fluorescent imaging- shining a light on the tracers which then light up as well. However, this doesn’t necessarily mean we see how they fight. The signal that comes from the labeled mAbs in the animals could be actually away from the sites of action when the mAbs are not bound to the targets at all. Therefore a technology that directly shows the mAb-target engagement is highly desired.
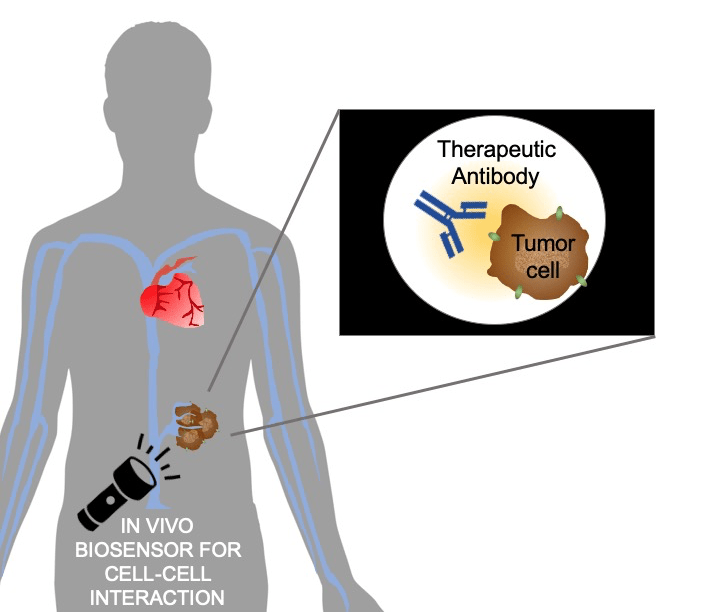
In our recently published study, we applied a bioluminescence resonance energy transfer (BRET) technology for directly visualizing mAb-target engagement in living animals. Bioluminescence is a natural process by which some living organisms like fireflies and jellyfishes produce light. In a BRET system, a bioluminescent energy donor and a fluorescent energy acceptor are usually included. Fluorescence is another process by which substances can produce light after absorbing light or other forms of energy. While fluorescent energy acceptors are the fluorophores, bioluminescent donors are usually luciferase, an enzyme that exists in many animals, such as fireflies.
Notably, the energy transfer between donor and acceptor is highly dependent on the distance between them. It’s like the energy donor and acceptor are playing fetch catch. If they are too far from each other, one can’t throw the ball to another one. When the energy donor is close to acceptor, the bioluminescent energy will be transferred from donor to acceptor. When the donor and acceptor are far from each other, the energy cannot be transferred and only bioluminescence can be detected in the system. Because of this proximity-dependent property, BRET technology is frequently used to identify protein-protein interaction. In Tang et al., NanoLuc, a small luciferase, was fused to EGFR. Anti-EGFR antibody cetuximab was labeled with a fluorophore. Upon cetuximab-EGFR binding, the fluorophore on cetuximab emitted light, directly demonstrating the proximity of mAb to the target, suggesting their engagement.
Our study showed some interesting properties of mAb-target engagement in lab animals with human tumors. First of all, the mAb-target binding did not line up with plasma cetuximab concentration. The binding usually came later in time than the peak of cetuximab concentration in the blood plasma. The delay between plasma concentration and binding in tumors was likely caused by the slow distribution of the mAbs from blood to the tumors, after injecting in the blood. Secondly, even at a very high dose, which is five times higher than the therapeutic dose, the disease targets in the tumor were not all engaged with cetuximab. This finding suggests a possible saturation of antibody penetration into the tumor. Even though we give patients high doses of mAb, it is possible that parts of the administered mAbs are not even helpful. This can happen when mAbs bind to the antigens at regions adjacent to blood vessels, preventing mAbs to further penetrate in the tumor.
Overall, in our study, we see how a mAb hug their targets in solid tumors, for the first time. This study demonstrated that BRET-based imaging methods amenable to determine mAb-target interactions in both space and time on living animals, further bolstering the applicability of BRET-based target engagement observation method in animals, broadly toward clinically viable and attractive targeted therapies. If simply elevating the doses of mAbs will not help patients to achieve better therapeutic effects, better dosing strategies or delivery methods should be considered.
Edited by Manisit Das
Work Discussed
Tang, Y., Parag-Sharma, K., Amelio, A. L., & Cao, Y. (2019). A Bioluminescence Resonance Energy Transfer-Based Approach for Determining Antibody-Receptor Occupancy In Vivo. iScience, 15, 439-451.

Leave a comment