Reading time: 4 minutes
Manisit Das
From the early days of OncoBites, we are trying to share with you the reasons we get cancer. Cancer arises when the DNA in our cells is altered due to irreparable mistakes in the cellular machinery or environmental effects. If a mistake happens in the proteins that help repair our DNA, or perform crucial functions in the growth and survival of cells, the consequences can be disastrous. Cells continue to accumulate more mistakes, grow uncontrollably, drain resources from healthy cells, and develop tumors.
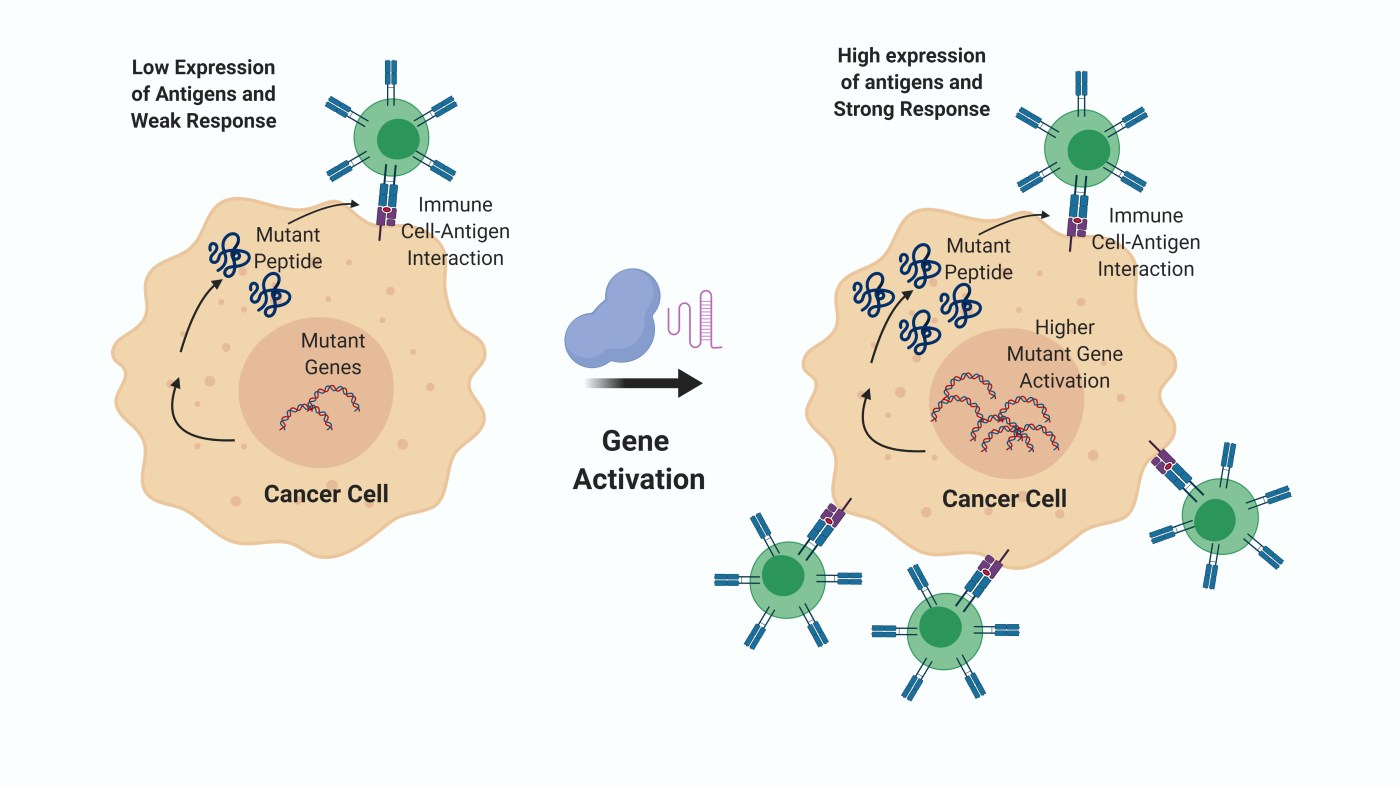
Cancers can be recognized, targeted, and eliminated by our immune system. Our immune cells can see a mutated or abnormally expressed protein that they are not trained to recognize as ‘self’ and destroy the cells that express them. However, cancers have a few tricks up their sleeves. Normally, all cells routinely have to present fragments of their protein contents (aka antigens) on the cell surface for review by immune cells. When some of these immune cells, such as T cells, sense a protein fragment as non-self (a viral antigen, or a protein fragment containing a mutation that is not seen wildly), they can destroy the target cell expressing the rogue antigen.
Cancer cells can suppress the expression of protein fragments on their cell surface, to evade an immune attack. Recently, a group of researchers from Yale University published a study where they ask if you can boost the expression of tumor-associated antigens to restore antitumor immunity, destroying cancer cells? By using CRISPR activation (CRISPRa) – a cousin of the more famous gene-editing method CRISPR interference (CRISPRi) which exploits an enzyme called Cas9 and evolutionarily conserved DNA motifs that are part of bacterial immune defense, and subsequently engineered by humans to edit genes, they increased the level of antigen expression on cancer cells. CRISPRa uses a modified version of the Cas9 enzyme, allowing scientists to increase, instead of suppressing gene expression as performed using CRISPRi. This resulted in the elevated presentation of antigens on the cell surface, improved T cell recognition, and finally, higher cancer cell killing. In animal models of cancer, they injected mice with cancer cells engineered via CRISPRa to express high levels of antigen, in animals either having existing tumors or with no existing tumors. This study was aimed at understanding whether this approach can be used in both preventive as well as therapeutic scenarios. In all these experiments, the cells were treated with a drug to arrest their capacity to divide, thereby preventing the engineered cancer cells from forming tumors. When used in a preventive situation, i.e., in animals with no existing tumors, the animals that received the cell treatment rejected tumors when challenged with cancer cells, in 100% of the cases. In a therapeutic scenario, the animals that received the cells as a treatment had partial shrinking of the existing tumors. The efficiency of the therapy was significantly improved when combined with an immune checkpoint inhibitor– therapies that allow T cells to perform better against cancer cells.
In the final part of the work, the authors expand the capability of their approach in treating existing tumors. They use Adeno-associated viruses as a vector to locally introduce genes in tumor cells on the model animals, thereby lowering the potential of off-target gene expression. The authors further demonstrate that they can use this therapeutic approach precisely optimized to each patient’s tumor by analyzing mutations in their tumors and boosting the expression of those unique mutant protein fragments, triggering an antitumor immune response.
Although the results are promising based on the preclinical study, we need to exercise caution. Gene editing technologies such as CRISPR are still in its formative stage when it comes to clinical trials. Although CRISPR-based gene-editing trials are slowly making it to humans, we are yet to fully learn the risks of such applications. Before this therapeutic approach moves to the clinical stage, it needs to be ensured that the gene-editing will not result in any significant off-target gene expression, giving rise to secondary cancers, or immune-related adverse events. However, this study is unique and shows a new direction in cancer immunotherapy. By allowing tumor cells to be seen better by the immune cells, we see a robust antitumor response in preclinical models of cancer, in an encouraging find, warranting follow-up studies in human patients.
Edited by Nisitha Sengottuvel
Work Discussed:
Wang, G., Chow, R. D., Bai, Z., Zhu, L., Errami, Y., Dai, X., . . . Chen, S. (2019). Multiplexed activation of endogenous genes by CRISPRa elicits potent antitumor immunity. Nat Immunol. doi: 10.1038/s41590-019-0500-4
Image created with Biorender

Leave a comment