Reading time: 4 minutes
Manisit Das
In the past, we have discussed checkpoint inhibitors – therapies that block communication between cancer and immune cells, preventing cancer cells from evading the immune system. Dr. James Allison and Dr. Tasuku Honjo, who won the Nobel Prize in Medicine in 2018 for their pioneering work in cancer immunotherapy independently studied the checkpoint molecules CTLA-4 and PD-1. PD-1, a molecule on immune T cells, can interact with PD-L1 on cancer cells and prevent the T cells from recognizing the cancer cells and launching an immune attack. Checkpoint inhibitor therapies capable of blocking PD-1, PD-L1, and CTLA-4 have been approved for treatment against several types of cancers.
Despite the improvement in therapies that can act as brakes to checkpoints like PD-L1, many patients will not respond to checkpoint inhibitors. The resistance to the therapies will also be guided by an increase in other checkpoints like LAG-3, which allow cancer to survive and escape the immune system, like the many-headed Hydra, which would grow two heads for every head that is chopped off. A current antibody therapy in clinical trials is trying to simultaneously brake PD-L1 and LAG-3, in the hope of achieving a more potent therapeutic benefit and bypassing resistance development.
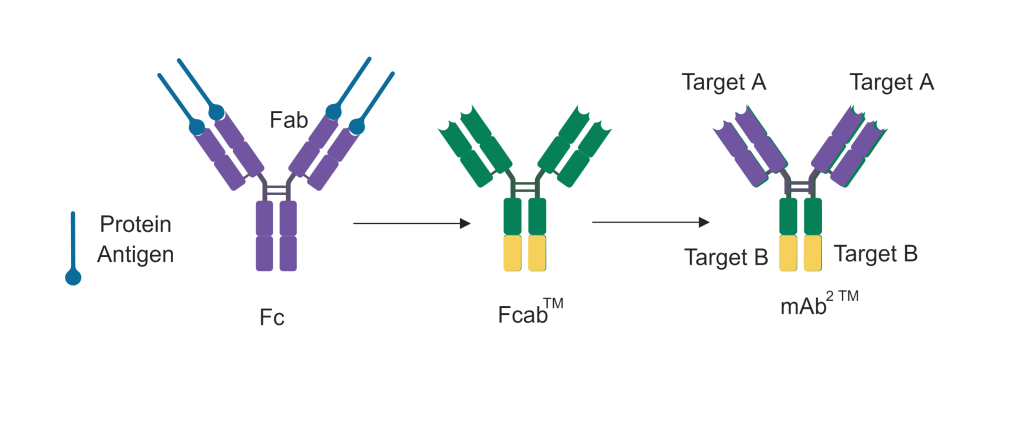
Antibodies are large (larger than an average protein in the human body) Y-shaped proteins that are used by the immune system to fight pathogens. Each antibody can recognize unique parts of proteins (and occasionally other molecules) on foreign substances called antigens. After this ‘match’—,the antigen-antibody recognition event— antibodies can directly neutralize the antigen and alert the immune system to the intruder carrying the antigens. In the arena of medical science, antibodies are often used to block proteins playing a critical role in diseases, including all the currently approved checkpoint inhibitor therapies against cancer.
Antibodies have two major regions; the Fab region, composed of variable sequences that allow it to recognize proteins with different sequences, and the Fc region, which communicates with the rest of the immune system sending signals after the antibody binds to a protein. Researchers have been playing with the basic design of antibodies to create potent new therapies for decades. A common approach involves engineering the arms of the antibodies, allowing it to recognize two different proteins. In 2010, a team of researchers from Vienna, Austria designed a new type of antibody called Fcab™ by engineering the Fc region, allowing the antibody to engage with an antigen through the Fc region as well. When this engineered Fc region is fused into a classical antibody, you get a new construct mAb2 ™ capable of binding to two different target proteins without compromising on the number of arms it can use to attach to the targets. These technologies are now a part of a company called F-star Biotechnology Ltd., and they are interested in using this approach to develop immunotherapies against cancer.
F-star is using mAb2 ™ to develop a checkpoint inhibitor capable of blocking both PD-L1 and LAG3. Recently, in a meeting of the American Association for Cancer Research (AACR) F-star showed data demonstrating the efficacy of their bispecific antibody against PD-L1 and LAG-3 in a mouse model of colon cancer. They also showed evidence of antibody target engagement in monkeys and a decrease in LAG-3 expression on T cells in mice. When treated with their bispecific antibody, T cells showed lower LAG-3 expression in comparison to the combination of single agents targeting PD-L1 and LAG-3 individually where LAG-3 expression was increased, suggesting the antibody’s potential to bypass LAG-3 mediated resistance of anti-PD-L1 therapy.
Currently, F-star is recruiting for a first-in-human early phase clinical trial with about 50 cancer patients who previously had progressive disease with PD-1/PD-L1 therapy. The goal at this stage is to determine the safety, tolerability, and activity of the PD-L1/LAG-3 bispecific antibody drug by 2020, and proceed accordingly with the testing of the drug.
F-star’s data suggest that their bispecific antibody drug is capable of evading the checkpoint-mediated immune escape of cancer. However, several questions remain. It is not clear if checkpoints other than LAG-3 like HAVCR2 (also known as TIM-3) will be increased after treatment with the bispecific antibody. Moreover, whether resistance mediated by LAG-3 expression is completely avoided or the onset of resistance is delayed, is not known. Further investigations will be necessary to answer these questions. Nevertheless, this drug is focused on treating an important targeted group of cancer patients, and it is exciting to see the drug’s transition to clinical testing. Whether the drug will make it to the market and if using two brakes is an effective strategy to stop cancer cells, time will tell, starting with the readouts of the Phase I trial potentially in 2020.
Works Discussed:
A LAG-3/PD-L1 mAb² can overcome PD-L1-mediated compensatory upregulation of LAG-3 induced by single-agent checkpoint blockade. Faroudi et al. (March 2019) – Poster at the annual AACR meeting
Wozniak-Knopp, G., Bartl, S., Bauer, A., Mostageer, M., Woisetschlager, M., Antes, B., . . . Ruker, F. (2010). Introducing antigen-binding sites in structural loops of immunoglobulin constant domains: Fc fragments with engineered HER2/neu-binding sites and antibody properties. Protein Eng Des Sel, 23(4), 289-297. doi: 10.1093/protein/gzq005
Image Credits:
Hercules and the Lernaean Hydra – Gustave Moreau
Antibody art created by the author with Biorender


Leave a comment