Reading time: 5 minutes
Diana Moreira
The need for new and improved anti-tumor therapies has led the research field to implement different strategies to reduce, and ideally cease, tumor development. With growing publications in the field of cancer metabolism in recent years, different tumor vulnerabilities are being uncovered, increasing the likelihood of finding new therapeutic targets.
This article will address in greater detail how the modulation of cancer metabolism can constitute a potential anti-tumor therapy. First it is important to address some key concepts in order to understand how metabolism can affect tumor growth and development.
The initiation and development of tumor cells results from the acquisition of multiple mutations over time, during a complex evolutionary process. These mutated cells become more prone to overcoming cell defense mechanisms and gaining limitless replicative potential. One of the main questions that arose among researchers in the field is how these cells are able to sustain their high proliferation rate. To answer this, some researchers began to explore the role of cell metabolism as a possible driver of cancer proliferation.
What is metabolism and why is important? Metabolism is essentially the breakdown of food/nutrients in order to obtain energy and other chemical compounds necessary for maintaining cell functionality. A large and complex network of different metabolic pathways coordinates this process at the cell/tissue level, where several enzymes and other molecules take the lead role. Using a simple analogy, the food/nutrients serve as fuels that feed different engines at the cell/tissue level, which contributes to an overall healthy state.
Tumor cells take advantage of this fine-tuned system to maintain their high proliferative state, increasing and altering the activity of some of these pathways and molecules due to their high demand for nutrients. These cells adopt a metabolic program in a process known as metabolic reprogramming. This process relies heavily on the formation of building blocks, known as nucleotides, lipids, and proteins that are essential for highly dividing cells. Fuels like glucose and amino acids feed these major metabolic pathways, which makes them attractive targets for dysregulating tumor metabolism and, consequently, tumor growth.
How is metabolic reprogramming first established and modulated over time?
Metabolic reprogramming in cancer is dependent on genetic and environmental cues. The acquisition of mutations over time can alter the genetic program of several components of metabolic pathways, including enzymes and transporters, which are critical for the import of nutrients into the cell. Apart from genetic factors, the tissue that surrounds and supports tumor cells shapes the molecular composition of the tumor microenvironment, which controls the availability of nutrients at the tumor site. The local acquisition of nutrients by different cells can impose a metabolic pressure in the tumor microenvironment forming a metabolic community at the tumor site. Herein, the exchange of metabolites, which constitutes the intermediate or end product of cell metabolism, can affect the metabolic pathways and the overall function of all the cells in this community.
The local availability of nutrients at a tumor site can be influenced by the overall amount of metabolites and nutrients that circulate in the bloodstream of an organism The systemic levels of metabolites along with their diversity reflect the composition of an organism’s diet. This interesting link between diet and cancer metabolism is now being explored as a potential tool to alter tumor metabolism through the modulation of dietary composition.
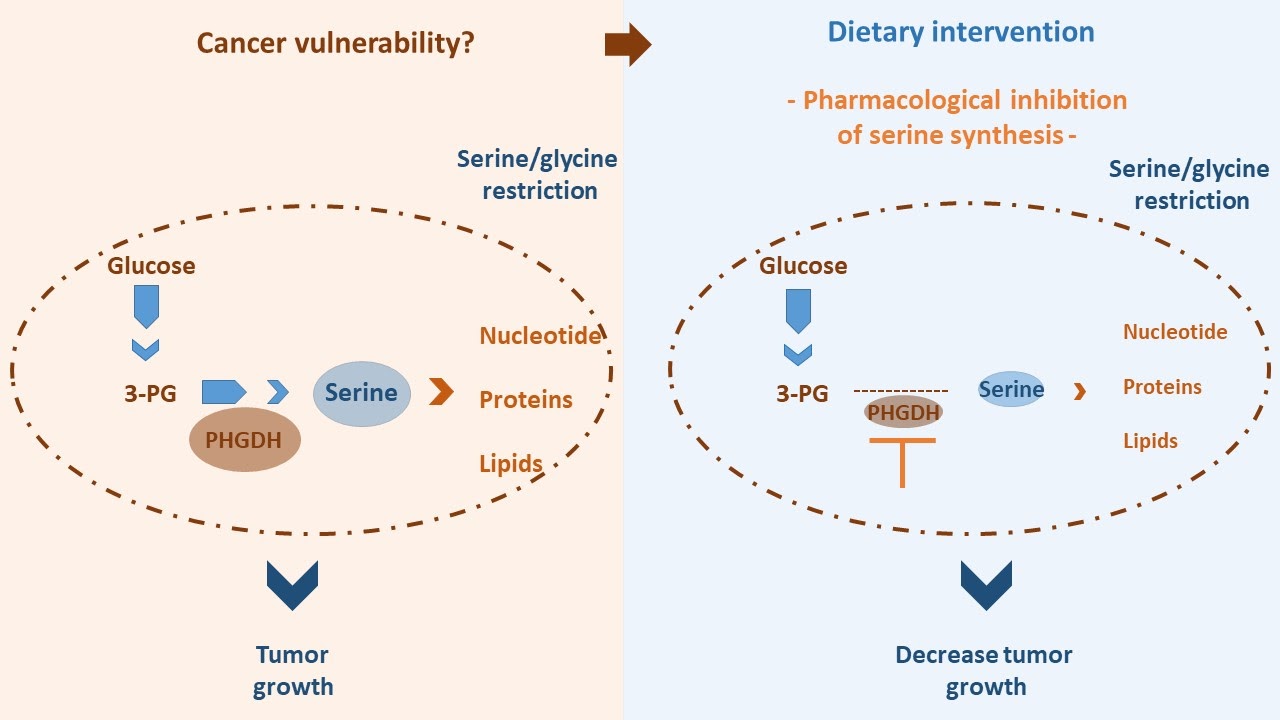
Cancer cells acquire metabolites in quantities that often exceed the levels of those acquired by healthy cells. This unique avidity for certain nutrients distinguishes a healthy cell from a mutated cell. Such metabolic features constitute the basis of recent dietary restriction strategies. In a study conducted by the lead author Bryan Ngo from Weill Cornell Medicine, was implemented a dietary strategy focused on the pharmacological depletion of nutrients. This study selectively restricted the production of nutrients like serine and derived glycine in mouse brain metastasis. These two amino acids have a broad effect in several metabolic processes such as the formation of nucleotides, lipids and proteins, just to name a few. In cancer cells, serine and glycine are essential amino acids. These cells are highly dependent on the import of these nutrients from the environment to maintain their highly proliferative state. In some types of cancers where a reduced level of serine and glycine is imposed by the local environment; the tumor cells need to produce serine by a process known as serine synthesis pathway to support tumor growth.
In the recent work from Ngo and colleagues, the authors found a restrictive environment for serine/glycine formed locally at mouse brain metastasis. This metabolic feature exposes the serine synthesis pathway as an important cancer vulnerability that could potentially be explored as an anti-tumor target.
The synthesis of serine from 3-phosphoglycerate, one of the steps of glucose degradation pathway (glycolysis), is highly controlled by the enzyme 3-phosphoglycerate dehydrogenase (PHGDH). This enzymatic reaction is central to support serine synthesis. In the same study, the pharmacologic inhibition of the enzyme PHGDH decreased brain metastasis and improved mice survival, without affecting extracranial tumor growth. This result highlights the dependency of brain metastasis on the serine synthesis pathway. Such a pharmacological strategy might have a potential therapeutic application, since the enzyme PHGDH was defined by the authors as a major driving factor of brain metastasis in different human cancer types.
Overall, the local restriction of nutrients described in this study induces cell metabolic reprogramming that selectively converts brain metastasis into a more attractive target for serine synthesis inhibition. This report highlights the major impact of cancer metabolic landscape (e.g. brain metastasis) for the outcome of potential dietary interventions.
Works discussed
Bryan Ngo, Eugenie Kim, Victoria Osorio-Vasquez, Sophia Doll, Sophia Bustraan et al. “Limited Environmental Serine and Glycine Confer Brain Metastasis Sensitivity to PHGDH Inhibition”. Cancer Discovery (2020). 10.1101/2020.03.03.974980
Christian Frezza. “Metabolism and cancer: the future is now.” British Journal of Cancer (2020). 10.1038/s41416-019-0667-3
Naama Kanarek, Boryana Petrova and David M. Sabatini. “Dietary modifications for enhanced cancer therapy.” Nature (2020). 10.1038/s41586-020-2124-0
Shree Bose, Annamarie E. Allen and Jason W. Locasale. “The Molecular Link from Diet to Cancer Cell Metabolism.” Molecular Cell (2020) 10.1016/j.molcel.2020.05.018
Edited by Kayla Snare

Leave a comment