Reading time: 6 minutes
Kate Secombe
Have you ever thought about how the cells in our body die? There are many ways a cell can die due to both external or internal factors, playing a role in keeping the body healthy and removing dangerous cells from the body system. When cells fail to listen to death signals, diseases such as cancer can occur. In fact, a tumor is a heap of unwanted cells that haven’t died as they should. A range of cell death pathways can release these death signals, and forcing these pathways is, therefore, a key facet of many anti-cancer therapies. This can be difficult though; cells can become resistant to these medications, or the anti-cancer therapy may kill many healthy cells, too. To overcome these issues, researchers are constantly developing new treatments that aim to more specifically kill cancer cells while minimising side-effects in other parts of the body. Oftentimes, these new treatments leverage atypical and not-as-readily activated cell death pathways, such as ferroptosis.
Ferroptosis, an iron-dependent cell death pathway, is one of the most newly discovered cell death pathways that has high potential to be useful in treating cancer. Cancer cells are known to have higher requirements of iron than normal cells, and current research suggests that inducing ferroptosis in cancer cells could cause cellular death in a way that other cancer treatments have been unable. In this article, we will look at what is known about internally-driven cell death pathways (known as programmed cell death), how ferroptosis is different, and how this process may be used to target a variety of cancer types.
Unpacking programmed cell death
Programmed cell death is an organised process where molecular signaling pathways cause the eventual breakdown of a cell. In multicellular organisms (like humans), a commonly activated pathway is known as apoptosis. In an adult human, about 60 billion cells die every day, with a great proportion of these dying via apoptosis! This is normal and is due to the continual replacement of old cells with new cells in places like the skin and intestinal lining. When a cell is triggered to undergo apoptosis, specialized proteins begin to break down cellular components and the cell’s DNA. While apoptosis is one of the more well-known forms of programmed cell death, there are many forms, and more newly discovered pathways are being widely researched currently.
Discovering ferroptosis
Ferroptosis, derived from the Latin word ferrum (iron), and the Greek word ptosis (a fall), is an iron-dependent form of programmed cell death. Ferroptosis was first fully described in 2012, however ferroptosis inducers were discovered as far back as 2003. These inducers were found to cause death of tumour cells differently to traditional apoptosis, but it was not until 2008 that it was determined that this type of cell death required iron to take place.
As well as being dependent on iron, ferroptosis is characterised by the accumulation of lipid peroxidases, enzymes that break down fats found in cellular membranes. This breakdown of the cellular membrane will continue as lipid peroxidase production continues, until, finally, resulting in cell death. Iron molecules are a key part of this process, as they are required to catalyze the lipid peroxidation process, and are also needed for other enzymes that help accumulate lipid peroxidases.
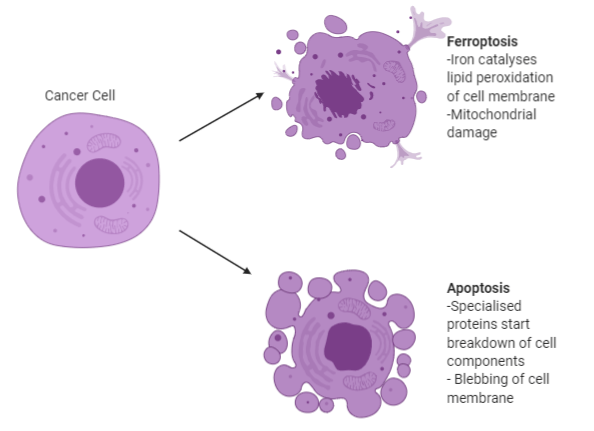
Ferroptosis is different from programmed cell death (such as the aforementioned apoptosis) in a variety of ways. From the outside, apoptosing cells appear to dissolve, whereas ferroptotic cells experience cell death more internally in the cell (mainly in the mitochondria) and there is a less obvious external phenotype of cell death (see figure 1 for more details). On a genetic level, the genes that regulate ferroptosis have no role in regulating any other form of cell death. These vast differences may prove useful in optimising cancer treatment in ways the use of apoptosis has been unable to.
Strengthening cancer therapies through iron-dependent cell death
Unfortunately, triggering apoptosis with anti-cancer treatments can have limited effectiveness in treating cancer, as many cancer cells demonstrate acquired or innate resistance to therapies that rely on apoptosis activation. When these therapies are avoided by cancer cells, this can impact survival and prognosis, and treatment plans will often need to be altered.
Ferroptosis has the potential to be far more effective in two key ways. Firstly, small amounts of evidence have led to speculation that ferroptosis may be able to reverse resistance to some forms of conventional chemotherapy via modulation of the enzyme glutathione peroxidase 4 (GPX4). GPX4 prevents lipid peroxidation and is one of the key regulators of ferroptosis. Particular types of therapy-resistant (also known as chemoresistant) breast cancer cells have been shown to be highly dependent on the GPX4 pathway to survive. A wide range of molecules have been developed to directly or indirectly inhibit GPX4 inhibit GPX4 or other molecules in its pathway, increasing tumor sensitivity to ferroptosis, and, ultimately, cancer cell death.
The second way ferroptosis has been demonstrated to be effective against cancer is in conjunction with immunotherapy. Immunotherapy is a growing area of cancer treatment where the patient’s own immune system is primed and targeted to help fight their cancer. This can be done in a few different ways, including removing barriers to the immune system attacking the cancer, such as in checkpoint inhibitors. This field is rapidly developing and new treatment paradigms are released nearly every day. It is beyond the scope of this article to explore the nuances of immunotherapy, but more information can be found on OncoBites here. In the immunotherapy field, there is research showing that ferroptosis may be useful in recruiting immune cells to the cancer site. Cells undergoing ferroptosis are known to release a molecule called HMGB1. This can lead to the release of cytokines and other immune cells involved in inflammatory reactions in the area. By releasing HMGB1, cancer cells undergoing ferroptosis, therefore, recruit immune cells to the tumour site. Further, a recent study also showed that specific immune cells (known as T cells) promoted ferroptosis, which led to better clinical outcomes. In this study, these particular T cells released a cytokine called interferon gamma, which was able to downregulate two subunits of the xc– system (which GPX4 is a part of), thereby promoting cell lipid peroxidation and subsequent ferroptosis.
Next steps
To date, much of ferroptotic research has been carried out in vitro (in laboratory grown cells as opposed to in animals or humans). Therefore, the obvious next step is to trial ferroptosis inducers in more sophisticated models. This research is currently underway, with one recent publication showing that a ferroptosis inducer known as IKE was effective in inhibiting the xc– system, and thereby slowing tumor growth in a mouse model of lymphoma. Another mouse model showed that the ferroptosis inducer erastin enhanced X-ray irradiation-mediated cell death, demonstrating that in that model, ferroptosis played a role in enhancing radiotherapy efficacy.
However, there are still some areas that need further research before large-scale clinical trials can occur. These include an understanding of any potential adverse effects of ferroptosis induction, understanding how the sensitivity to ferroptosis might change in different types of tumours, and understanding how lipid peroxidation is functionally linked to ferroptosis. In conclusion, inducing ferroptosis in cancer cells could be an exciting way to improve the specificity and longevity of a variety of cancer treatments.
Edited by Keighley Reisenauer
Key readings
Cotter TG. Apoptosis and cancer: the genesis of a research field. Nat Rev Cancer. 2009;9(7):501–507. doi:10.1038/nrc2663
Xu T, Ding W, Ji X, et al. Molecular mechanisms of ferroptosis and its role in cancer therapy. J Cell Mol Med. 2019;23(8):4900–4912. doi:10.1111/jcmm.14511
Hassannia B, Vandenabeele P, Vanden Berghe T. Targeting Ferroptosis to Iron Out Cancer. Cancer Cell. 2019;35(6):830–849. doi:10.1016/j.ccell.2019.04.002
Mou, Y., Wang, J., Wu, J. et al. Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J Hematol Oncol 12, 34 (2019). doi: 10.1186/s13045-019-0720-y