Reading time: 4 minutes
Manisit Das
Nanoparticles are tiny substances, about a hundred- to thousand-fold smaller than a typical cell, or about ten thousand-fold tinier than a grain of salt. In the past, we have discussed how nanoparticles are being investigated as carriers of drugs to tumors. These investigations were in part fueled by a common belief among scientists that blood vessels draining to solid tumors can get leaky, allowing nanoparticles to efficiently get inside the tumor, but not normal tissue with healthy blood vessels. However, there is limited evidence of whether such an ‘enhanced permeability and retention(EPR) effect’ exists in human cases of cancer and it is hotly debated whether the phenomenon is unique to model rodent tumors.
Over the last three decades, the EPR effect served as a logical basis for the field of nanomedicine. Research groups have performed several studies using nanoparticles as a drug delivery system, utilizing their ability to get through leaky vessels and to circulate for a longer time through blood when attached to specific polymers. Together, these features enabled superior tumor delivery of the drug, when compared to direct injection in a laboratory setting. The end goal was to reduce the side-effects of toxic chemotherapy drugs and increase therapeutic efficiency. The approach encountered a lot of setbacks while trying to translate approaches executed in research laboratories to the clinic. Nevertheless, we saw the approval of several nanoparticle-based drug formulations in the past twenty-five years, ranging from Doxil in 1995 for AIDS-related Kaposi’s sarcoma to the more recent Onivyde for metastatic pancreatic cancer in 2015.
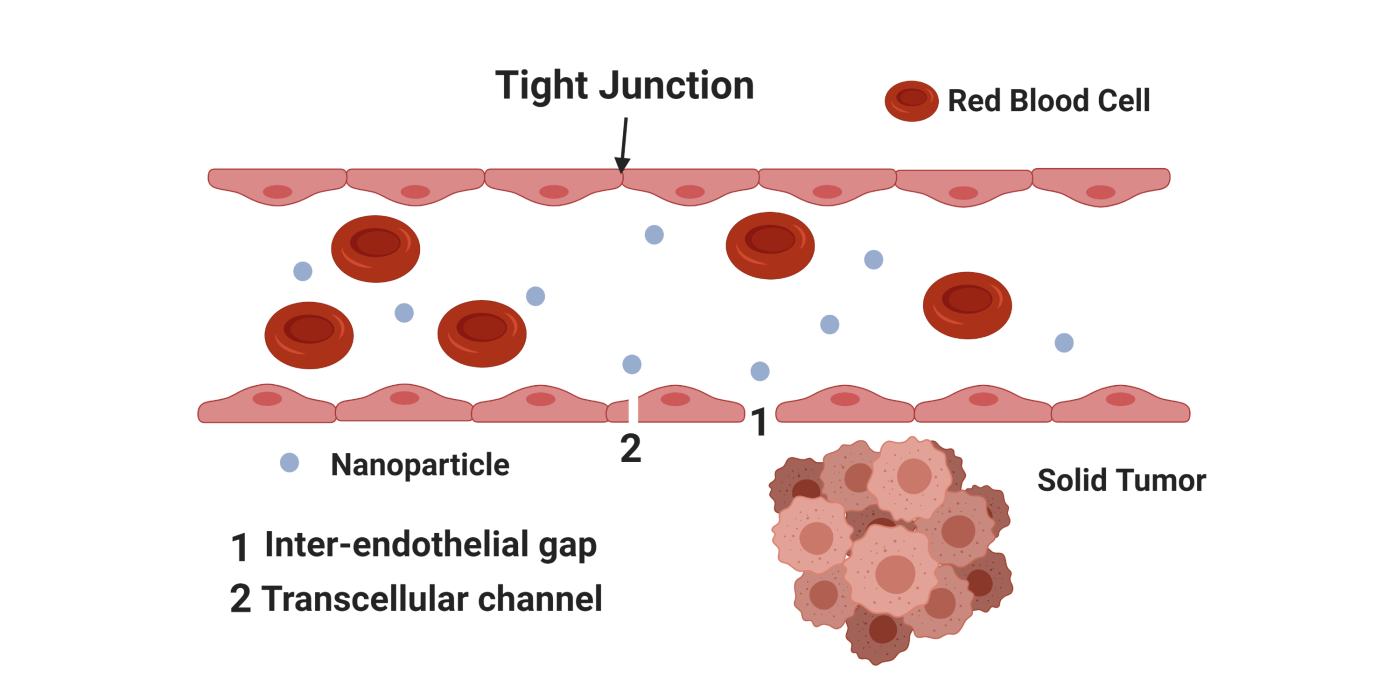
A recent study performed by a team of researchers based in Toronto raises important questions about the mechanism of nanoparticle entry inside solid tumors. Using microscopy, mathematical models, and distribution studies in rodents, they found that within the layer of endothelial cells that line the blood vessels, there aren’t enough gaps that can explain the actual accumulation of nanoparticles inside the tumor. They found that the gaps between the endothelial cells, also known as inter-endothelial gaps, covered only about one in two thousand parts of the blood vessel surface area. This low number couldn’t account for all the nanoparticles that could get into the tumor when fitted into the mathematical equations.

Using microscopy, the researchers also found features that hinted at the transport of materials through the reorganization of the cellular structure, called trans-endothelial transport. Cells often use these transport pathways to get nutrients inside cells and move them from one cell to another. In order to understand if this is a major pathway for nanoparticles to get inside tumors, they used a model that distinguishes between contributions from the active trans-endothelial mode of transport (requiring direct cellular activity, or “through the cells”) and passive modes of transport (through the gaps between the cells or “between the cells”). They found the gaps contribute to only about 3% of particle accumulation for small nanoparticles (about one-tenth of the size of a virus), however, that increases to about 25% when nanoparticles are as big as a virus.
These new results about the contribution of the gaps between cells to nanoparticle entry are questioning the premise of the original drug delivery concept, where researchers believed using larger nanoparticles can reduce drug accumulation in healthy tissues while enriching the deposition in leaky tumor tissues. It is concerning if delivery through the gap contributes to only about 25% of the nanoparticle accumulation, and the rest is accounted for by other mechanisms of transport such as active transcellular pathways. When the team looked into the patient tumor samples stored in Ontario Tumor Bank, they found the vessel architecture was similar to what was observed in mouse tumors- rare gaps and structural features of the cells hinting at active trans-endothelial transport.
So why do we care how nanoparticles enter into a tumor? A precise knowledge of the transport processes allows us to understand better how we can tweak the system to enrich drug delivery against cancer further, and improve the clinical translation of approaches that are being tested in the lab. For years, we had been minding the gap. Is it time to pay attention elsewhere? What does this mean for the future of nanomedicine in cancer therapy? Time will tell.
Edited by Gabrielle Jean Dardis
Work Discussed:
Sindhwani, S., Syed, A.M., Ngai, J. et al. The entry of nanoparticles into solid tumours. Nat. Mater. (2020) doi:10.1038/s41563-019-0566-2
Image created with Biorender

Leave a comment