Reading time: 5 minutes
Payal Yokota
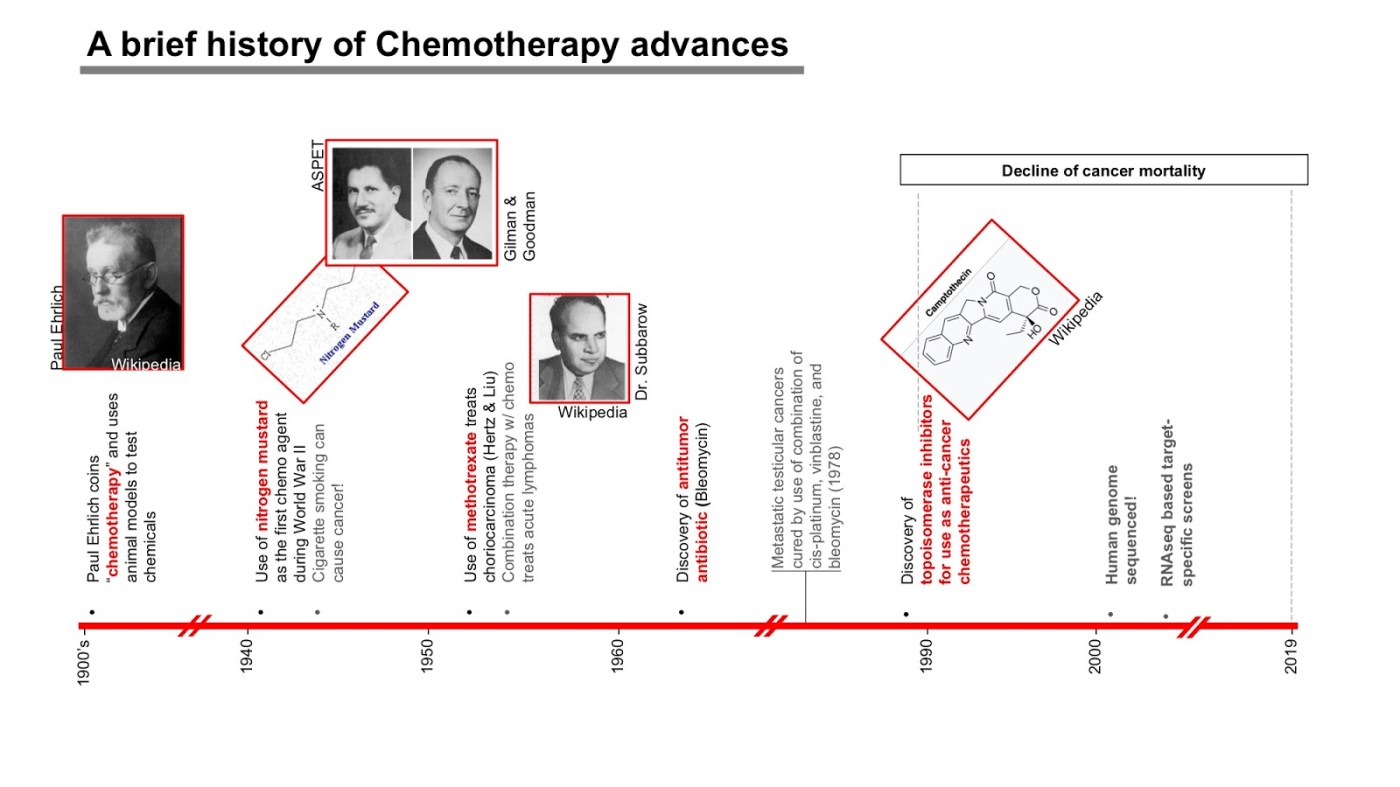
Chemotherapy (chemo) can be prescribed alone and in combination with other therapies such as surgery, radiation, and immunotherapies (ie. vaccines, checkpoint blockade, etc). Since its discovery, the chemo field has greatly advanced, both in its development and ability to treat cancers. While surgery and radiation act locally to remove a cancer from specific regions in the body, chemo can flow through the bloodstream and reach multiple niches throughout the body where cancer has spread. This works especially well to kill cancers that have spread across tissues and are difficult to remove surgically. This article will present a brief historical perspective on evolution of chemotherapy since its discovery, how its use can be expanded beyond a toxic cancer killing compound and examples of how that occurs. Figure-1 shows an at a glance view of the chronological account of pivotal discoveries in chemotherapy field.
To understand how chemo works to treat a large number of cancers, it is first important to understand what cancer really is. When an infection occurs via viruses or bacteria, the immune system sends signals to attack the infected cells and kill them. Unlike these foreign invaders, cancer is essentially a self-cell gone rogue, and therefore difficult to detect. In order to evade cancer, one must look for minute differences within the cancer cells that separate them from healthy or normal cells in the body. One striking characteristic of cancer cells is that they divide often and rapidly, which can be exploited for cell detection and ultimately stopping their division.
Discovery of chemo agents, 1940’s
In 1900, Nobel Laureate Paul Ehrlich suggested that there may be a way to fight microbes without harming the body. He called this concept the magic bullet. This idea of finding a chemical compound or treatment that is able to heal the body without causing detrimental damage was adopted by the field of cancer therapy four decades later. During World War II, Gilman and Goodman were recruited to evaluate potential applications of a lethal chemical called nitrogen mustard (Gilman et al, 1946). The initial studies were carried out in mice that were injected with human lymphoma cancer cells, then treated with nitrogen mustard. The chemical eradicated cancer by binding to a dividing cell’s DNA strand, which induced a signal for cell death. While this is an effective way of killing rapidly dividing cancer cells, it is risky because it also eliminates some fast dividing healthy cells, such as growing fetus during pregnancy or hair growth. As a result, patients treated with chemo often experience temporary hair loss in addition to nausea and fatigue.
Anti metabolites and microtubule disruptors, 1950’s
Towards 1950’s, two kinds of chemo drugs that induced cancer cell death by acting on specific phase of cell cycles were discovered. Unlike alkylating agents like nitrogen mustard that kill nonspecifically, these drugs fell in the categories of anti-metabolites (ex. methotrexate, synthesized by Dr. Subbaraw) or anti-microtubule agents (ex. paclitaxel or taxol) .oth anti-metabolites and anti-microtubules are heavily used for treating a variety of cancers. Anti-metabolites inhibit cell division by preventing conversion of folic acid into tetrahydrofolate, thereby preventing DNA synthesis in the S-phase of cell cycle (Newton H, 2012). In contrast, an anti-microtubule agent acts by disrupting the dynamics of mitotic spindles during G1 phase of the cell division cycle, thereby blocking cells from dividing normally and rapidly (Long and Fairchild, 1994).
Antitumor antibiotics, 1960-80’s
In the 1960s-80s, researchers discovered a special class of antitumor antibiotics. These are different from the antibiotics used to treat bacterial infections. Antitumor antibiotics interfere with the DNA inside of a cancer cell, thereby, slowing or stopping them from multiplying. These drugs are derived from natural products from soil fungus.
Enzyme inhibitors, 1980-90’s.
Another class of chemo drugs inhibit essential enzymes required for the replication machinery,resulting in the cell death of cancer cells. By the 1960’s, methotrexate was able to eradicate a specific type of uterine cancer. During the next two very productive decades sophisticated culture systems were developed to grow cancer cells and then implant human cancers into mice (xenografts), in order to scrutinize the molecular mechanisms that drive cancer progression. In the late 80’s and 90’s, a new class of chemo agents were introduced that blocked the replication machinery by acting negatively on enzymes known as topoisomerases. These drugs were different from previously described drugs, because these were small molecules that were able to efficiently stop cell replication of cancer cells.
Where are we now?
It’s been over seven decades since the original discovery of chemotherapy. Since then, patients diagnosed with solid and blood cancers have been treated with numerous formulations used in combination with surgery, radiation, and immunotherapies. However, chemotherapy continues to be used as a poison as it efficiently kills cancer and shrinks tumors either as the primary therapy or in combination with other approaches. Recent studies have suggested that chemotherapy at smaller doses can work to not only kill cancer cells, but also enhance the responses of the immune cells that usually efficiently kill infected and cancer ridden cells. A study published in Science Translational Medicine showed that chemo worked well in a group of patients with advanced cervical cancer; despite HPV vaccination, cancer developed in these patients. Chemo was able to kill cancer cells and some group of innate immune cells, but not killer T cells, which helped patients fight cancer better and faster (Welters et al., 2016).
Testing these hypotheses in animal models with different types of cancers can shed light on the efficiency of chemotherapy to kill cancer cells as well as activate essential compartments of the immune system. The reason each cancer patient responds differently to therapies is due to their personal immunological history and their microbiome, which is made up of good bacteria that help heal the lining of tissues after therapy. Genome sequencing has greatly improved our ability to predict what genes make our bodies vulnerable to developing cancers, thereby making predictions on best treatments for different cancers. We live in a golden era of cancer therapies and understanding older forms of therapy, such as chemotherapy, can help us design better solutions for individual patients such that the patient is treated without compromising quality of life.
Works Discussed:
Gilman and Goodman, Nitrogen mustard therapy. Use of methyl-bis(beta-chloroethyl)amine hydrochloride and tris(beta-chloroethyl)amine hydrochloride for Hodgkin’s disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. JAMA. 1946, 132:p126-132.
Newton H, Neurological complications of chemotherapy to the central nervous system
Handbook of Clinical Neurology, 2012. Vol. 105: p903-916.
Long BH and Fairchild CH. Paclitaxel inhibits progression of mitotic cells to G1 phase by interference with spindle formation without affecting other microtubule functions during anaphase and telephase, Cancer Research. 1994 Aug 15;54(16):4355-61.
Welters et al., Vaccination during myeloid cell depletion by cancer chemotherapy fosters robust T cell responses. Sci Trans Med., 2016, Vol 8 (334): 334ra52
DeVita VT and Chu E. History of Cancer Chemotherapy. Cancer Research – AACR. 2008; 68: (21) Nov 1.