Reading time: 4 minutes
Shaye Hagler
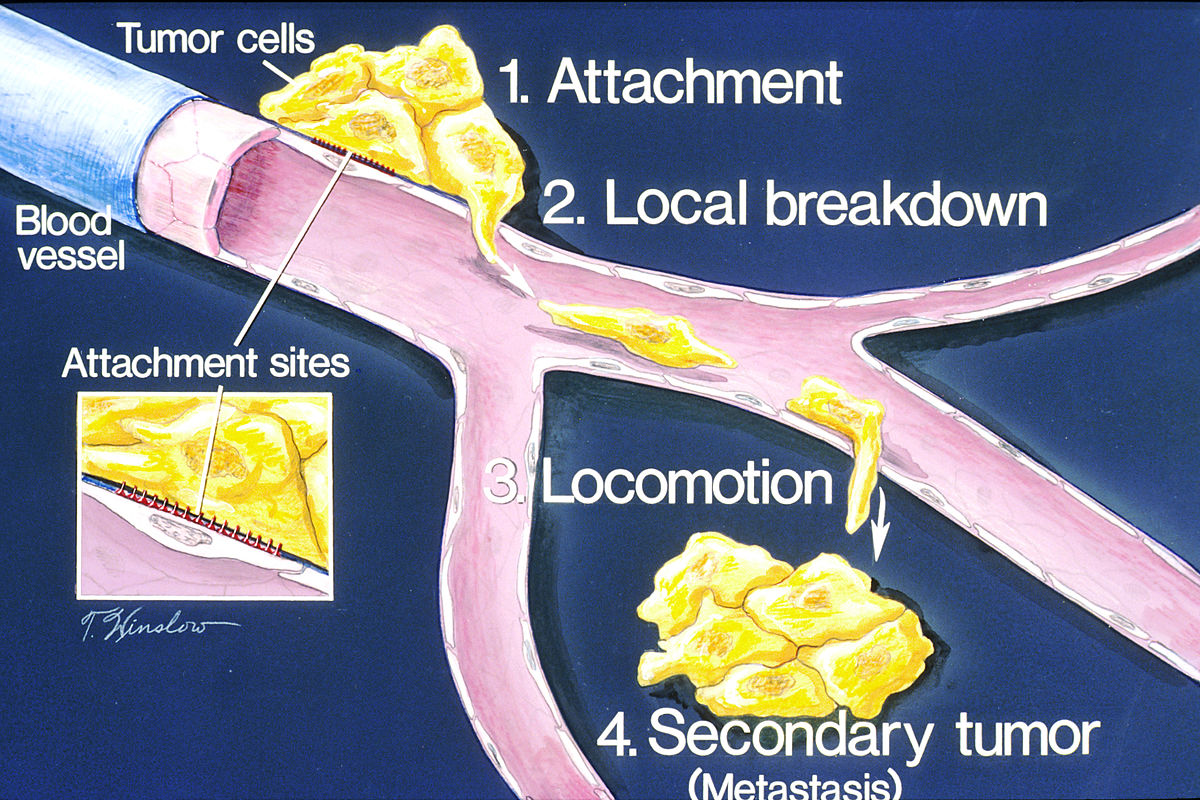
For those of you whose lives have been impacted by cancer, you might have heard of the term “metastasis.” We call cancer metastatic when it breaks off from the initial tumor and travels to other organs. You might also have heard it being referred to before as “stage 4 cancer.” Once cancer reaches this stage it is incredibly difficult to treat since it becomes much harder to track down tiny pockets of cancer cells and remove them via surgery. Therefore, metastasis is responsible for around 90% of cancer deaths. Last fall, fellow Oncobites author Rachel taught us about a few studies that showed us what triggers cancer cells to metastasize in the first place. This type of work is essential if we are going to learn how to stop cancer from metastasizing, but another preventative avenue we can explore is what makes certain organs more enticing for metastatic cells. For a long time, we’ve used the “seed-and-soil hypothesis” to explain this phenomenon. This hypothesis states that cancer cells can be thought of like seeds; just because they get spread elsewhere doesn’t necessarily mean they will be able to put down roots and grow. They need to end up in the right soil, which we can refer to as a pro-metastatic microenvironment (which just means we are talking about environment at a cellular level). The liver, being an extremely common destination for metastatic cells, is a good place to start asking: what makes the soil “right”?

Researchers at the Abramson Cancer Center of the University of Pennsylvania set out to answer this question using mice with pancreatic cancer. They removed spontaneous tumors formed in mice, made them express a yellow fluorescent protein so each cell could be easily tracked, and then re-implanted cells in the pancreas of mice that were either healthy or genetically altered to be more susceptible to cancer. They found that the latter group ended up with 3 times more liver metastases than healthy mice. In order to determine what genes may be involved in this process, they performed RNA sequencing on livers from both sets of mice. This revealed that the mice with higher rates of metastasis also had a specific immune reaction. When they dug into this reaction further, they discovered it was triggered by healthy cells near or within the original tumor that produce a protein called IL-6. IL-6 then travels through the bloodstream toward the liver, where it binds to a receptor on the outside of the liver cell. From there, a protein called Jak adds a molecule to the receptor that turns on another protein called STAT3 (this is just so much easier to see in a picture). Now, STAT3 is important because it’s what’s called a transcription factor; it’s a protein that binds to specific parts of DNA and triggers the cell make those sections into more proteins. In this case, researchers found that STAT3 was going on to drive the production of a protein called SAA.
I know that I just threw a lot of fancy protein names at you. The main point is: IL-6 leaves the original tumor, travels to the liver, and then starts a cascade of events that leads to increased production of SAA. SAA then goes on to change the microenvironment of the liver, recruiting immune cells, enriching the blood supply, and adding proteins that support extra cell growth. This is a major part of how the “soil” is changed and sensitized to the growth of cancer “seeds.” It’s pretty cool how these researchers were able to reveal this process too! They saw that the immune reaction seemed important, so they tested this cancer type in different mice where they could remove specific genes within the reaction pathway and see how that affected metastasis. When IL-6 was removed, SAA was decreased and metastasis occurred less often. When STAT3 was removed, no SAA was produced and metastasis occurred less often. What does the difference in SAA levels between the two experiments tell you? Answer: IL-6 is not the only activator of STAT3! All these data together tell us how liver cells are specifically involved in spreading cancer for the first time, and may lead us to examine preventative treatment for patients targeting the liver so that the “seeds” aren’t nurtured by healthy “soil” in the future.
Work Discussed:
Lee, J. W., Stone, M. L., Porrett, P. M., Thomas, S. K., Komar, C. A., Li, J. H., … & Black, T. A. (2019). Hepatocytes direct the formation of a pro-metastatic niche in the liver. Nature
Image Credits:
https://commons.wikimedia.org/wiki/File:How_metastasis_occurs_illustration.jpg
https://www.oklahomack.com/conditions-treated/metastatic-cancer
https://commons.wikimedia.org/wiki/File:Transcription_Factors.svg

Leave a comment