Reading time: 4 minutes
Morgan McSweeney
A research team from UC Berkeley, UCSF, and UNC Chapel Hill has developed a new strategy to prevent unwanted side effects of toxic chemotherapy drugs. Often, choosing a dose of chemotherapy has to be balanced between how effective it will be at killing tumor cells and how toxic it will be to the rest of the body. Unfortunately, many of the most effective cancer therapies are still relatively non-selective, and toxicities can stand in the way of continuing to provide therapy.
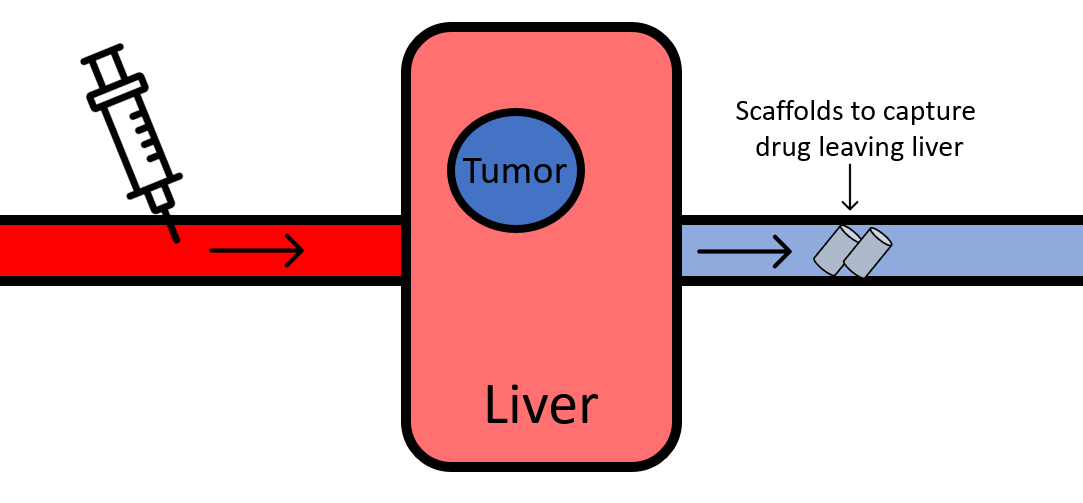
Patients who have certain types of liver cancer are sometimes treated with an intra-arterial injection of a drug called doxorubicin, as part of a strategy called transarterial chemoembolization (TACE). In patients with intermediate stage liver cancer, the use of TACE has improved typical survival from 16 months to 20 months. In the past, chemo drugs were usually administered into the patient’s veins. From there, it would circulate through the entire body until it made its way to the liver. More recently, clinicians have begun to test whether they can administer drugs directly into the artery that supplies blood to the liver. Theoretically, this strategy means that the drug does not have a chance to accidentally become stuck in other organs before it has a chance at getting to the liver tumor. However, even with this method, not all of the drug stays in the liver on its first pass, and the amount that escapes through the vein leading out of the liver can wreak havoc on the rest of the body. For example, in early studies of the use of doxorubicin for liver cancer conducted in the 1980s, up to 25% of patients who received doxorubicin died of fatal complications such as cardiotoxicity or septicemia.
To address this “escaped” fraction of drug, Oh et al. have designed a method that is able to soak up the molecules as they make their way out of the other side of the liver. They designed a 3D printed cylindrical scaffold that fits inside the vein leading out of the liver (see diagram below). This custom scaffold is coated with a chemical that binds tightly to the drug doxorubicin. In an animal model of the treatment setup, the researchers first placed the scaffold in the vein leading out of the liver. Then, they administered doxorubicin through the arteries supplying the liver, similar to current clinical practice. They compared how much drug made its way out to the rest of the body in tests where the scaffold was coated with the doxorubicin-catching chemical, versus uncoated control scaffolds.

The researchers found that animals that received the chemical-coated scaffold leaked ~64% less doxorubicin out to the rest of the body, compared to the control scaffolds. If this technique works similarly in humans, this could be a major therapeutic advantage. If, when using the drug-soaking scaffold, only one third as much doxorubicin can go cause toxicity to the rest of the body, that means that patients might be able to use up to 3 times more dox, improving the payload delivered to the tumor while maintaining the same level of safety currently achieved without any scaffold.
Although impressive for this model of liver cancer, this strategy could theoretically work well in any cancer that is restricted to an organ, and for which there is one or just a few veins through which blood leaves the organ. When targeting different organs, the 3D-printed nature of the scaffold means that this strategy could be adapted to match different blood vessel sizes. The scaffolds could even be adapted to individual patients using images from CT scan or MRI data. This would likely make each scaffold more expensive compared to a mass-produced, one-size-fits-all approach, but the extra customization could lead to better efficacy. The research team believes that, although this was an impressive first set of results, there could be room for further improvement. By changing the number of scaffolds placed in the vein, changing the size of the holes in the scaffold, or by changing the thickness of the drug-absorbing layer of coating, they hope to produce even better outcomes.
Work Discussed:
Oh, H. J., Aboian, M. S., Yi, M. Y. J., Maslyn, J. A., Loo, W. S., Jiang, X., . . . Balsara, N. P. (2019). 3D Printed Absorber for Capturing Chemotherapy Drugs before They Spread through the Body. ACS Central Science. doi: 10.1021/acscentsci.8b00700