Sara Musetti
Historians love to say that those who do not know their history are doomed to repeat it. In science, failing to remember and understand our history means that we may need to make the same discoveries again and again. This appears to be the case for cancer immunotherapy, a new branch of research that seeks to stimulate the immune system to recognize cancer cells as a threat and kill cancer. Immunotherapy is a buzzword in cancer research, delivering startling results in a small subset of patients. Scientists and doctors are scrambling to learn more and to expand the pool of patients helped by these therapies by developing new treatments and combinations.
However.
It turns out that immunotherapy, although it appears to have burst onto the scene with the approval of ipilimumab (trade name Yervoy) in 2011, is not a new concept at all. Ancient Egyptians documented the fact that tumors could be reduced in people with infections. German doctors in the 1860s-1880s found that patients with certain severe bacterial infections also experienced tumor shrinkage. They even went so far as to purposefully inject patients with bacteria in order to prove a causal effect. In 1891, Dr. William Coley brought immunotherapy to American hospitals; he developed his own special blend of bacteria, known as Coley’s Toxins, that he injected into patients with inoperable tumors over the span of the next forty years at Memorial Hospital in New York City. However, even with his successes, the scientific community failed to believe his results and the US FDA eventually shut him down. It would be decades before scientists in laboratories were able to explain why Dr. Coley’s unorthodox procedure worked, and could begin developing more controlled and targeted immunotherapies to help patients today.
Immunotherapy can take a lot of forms, from checkpoint inhibitors to CAR-T therapy that Liz discussed last week. One of the forerunners in cancer immunotherapy is known as a cancer vaccine. Most vaccines prevent against viral and bacterial infections. However, the broad definition of a vaccine is simply an agent that stimulates an immune response against a particular disease causing agent, including cancer cells. This is also why injecting patients with a bacteria can help shrink tumors; infections stimulate the immune system to be on high-alert for threats, and occasionally, the immune reaction to an infection will spread to recognize tumors and eradicate tumor cells. Essentially, Dr. Coley’s toxin injections acted as a rudimentary vaccine. One modern vaccine known to prevent cancer is Gardasil, which was approved in 2006, and protects against four
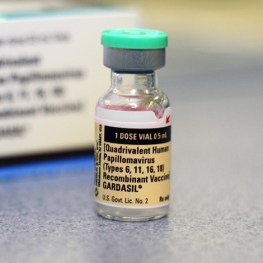
strains of human papilloma virus that are responsible for the majority of cervical cancers. Similar to a traditional vaccine, Gardasil protects against a virus that is linked to cancer initiation. Most cancers are not linked to a viral infection, but that doesn’t necessarily mean we can’t use vaccinations to treat them. Researchers are currently working on developing vaccines to treat cancer after it has been established, rather than to prevent cancer. In order to make a vaccine, you have to have a small part of the disease causing agent (bacteria, traditionally, or tumors in a cancer vaccine) that the immune system will recognize, this is called an antigen. In cancer, these antigens arise from mutations in normal proteins, these new antigens are called neoantigens. However, every patient’s cancer has unique mutations, and even within a single patient cancer cells can have different mutations. Therefore, each patient has a unique set of neoantigens, so there will never be a “one size fits all” vaccine for cancer therapy. However, it may be possible to characterize individual tumors for mutations and develop personalized medicine, such as vaccines tailored to each patient.

Recently, a pilot clinical study of 25 patients was conducted to test personalized cancer vaccines against ovarian cancer. This study focused on dendritic cells, which serve the purpose of presenting antigens to T cells, that attack diseased tissue. For this study, dendritic cells were harvested from patient’s blood and stimulated with mashed up tumor tissue in the laboratory. This tumor tissue contains the antigens that dendritic cells can use to teach T cells to recognize and kill tumors. This process allows the dendritic cells to take up and process neoantigens from the tumor tissue. The dendritic cells are then collected and injected back into the patient’s lymph nodes, the areas of the body where immune cells interact and coordinate a robust immune response. T cells travel from the lymph node in search of the antigens/mutations they’ve been trained to recognize and eliminate those threats. In most patients, this vaccine was combined with standard of care treatments: bevacizumab, a targeted therapy against growth factors that feed cancer, and cyclophosphamide, an immunosuppressive chemotherapy agent.
This study found that these vaccines were safe in all 25 patients tested, and that patients treated with the vaccine plus standard therapy slowed disease progression and had a much better rate of overall survival (almost double!) than patients who historically did not receive the vaccine. They chalked this difference up to the increased number of antigen-specific T cells that could target and kill tumor cells, thanks to the dendritic cell vaccine.

While a vaccine based on a patient’s own cells and the mutations specific to their tumors does raise a robust immune response that is compatible with each individual’s immune system, widespread use of personalized vaccines will take time and further testing. For one thing, this is a preclinical trial, with only 25 people. If they take this vaccine into clinical trials, it can still take a decade before it gets approved, if it does; the majority of therapies that enter clinical trials do not get approved. And personalized vaccines that require the use of a patient’s own cells and tumors are labor-intensive. Hospitals would need special facilities and professionals to prepare these vaccines and ensure safety on a case-by-case basis, and then store these personalized vaccines for the duration of therapy. This would be expensive, at least while these types of vaccines are rare and hospitals lack the infrastructure for it. However, it is becoming a more common option every day; there are a few cell-based therapies currently on the market, such as CAR-T, which Liz discussed last week. In 2010, a dendritic cell-based therapy called Sipuleucel-T (trade name Provenge) was approved by the FDA for prostate cancer. Provenge works by activating dendritic cells with a prostate cancer-associated antigen, rather than patient-specific tumor proteins, as proposed in this study. At the time of approval, Provenge cost $93,000 and extended life expectancy by four months compared to patients who did not get the vaccine. However, even with these concerns, the current vaccine work is a very promising result for patients, and highlights the amazing cancer-fighting abilities of our own bodies and clinician’s ability to unlock those abilities. Advances made today pave the way for better therapies tomorrow, as science continues to learn and grow. It took decades to understand the work of Dr. Coley and begin applying it safely to patients, but now we have immunotherapy. As we work toward safe and effective cancer therapies, it is more important than ever to learn and to build upon the work that came before us, because it offers us unique understandings of the human body.
Image Credits
Featured image: Vaccine, Original Author: Nick Youngson
Image caption: Gardasil. © Wikipedia
Image caption: Vaccine-Based Immunotherapy. Dendritic cells (gray) stimulating T cells (pink) to recognize an antigen. © NIH Image Gallery, presented without modification.
Image caption: Dendritic cell therapy. © Simon Caulton via Wikipedia Commons.
Primary Work Cited
Tanyi, J. L., Bobisse, S., Ophir, E., Tuyaerts, S., Roberti, A., Genolet, R., Baumgartner, P., Stevenson, B.J., Iseli, C., Dangaj, D. & Czerniecki, B. (2018). Personalized cancer vaccine effectively mobilizes antitumor T cell immunity in ovarian cancer. Science translational medicine, 10(436).

