Reading time: 4 minutes
Florence Roan
In 2024, the US Food and Drug Administration (FDA) approved lifileucel for the treatment of advanced melanoma. Lifileucel’s approval marked dual milestones. It was the first tumor-infiltrating lymphocyte (TIL) therapy approved for use in cancer, and the first cellular therapy approved for a solid tumor. This new approach—using TILs to target solid tumors—opens the door to new treatment possibilities for patients with some of the most difficult-to-treat cancers.
Harnessing the Immune System to Fight Cancer
All currently approved cellular therapies for cancer use a patient’s own T cells (or T lymphocytes). As part of the body’s surveillance system, T cells have sensors called T-cell receptors (TCRs) that recognize antigens, which are cell- or microbe-associated markers. Each person has billions of T cells with unique TCRs. This gives the immune system an extraordinary ability to recognize a vast range of threats. When a T cell recognizes an antigen, it can become activated to kill a target or recruit help.
T cells from a patient’s blood can be genetically engineered to recognize specific cancer antigens, creating modified cells called chimeric antigen receptor T cells (CAR T cells). To date, six CAR T-cell therapies have been FDA-approved for certain blood cancers. These cancers include B-cell acute lymphoblastic leukemia, large B-cell lymphoma, mantle cell lymphoma, follicular lymphoma, chronic lymphocytic leukemia, and multiple myeloma. For patients with these diseases, CAR T cells have the potential to dramatically improve survival and quality of life.
However, designing CAR T-cell therapy for solid tumors remains challenging. Unlike blood cancers, solid tumors often have a diverse array of poorly characterized antigens, making it difficult to identify the most effective CAR T-cell targets. In addition, CAR T cells are derived from cells circulating in the blood, which have difficulty migrating to and persisting within solid tumors. By using T cells already in the tumor, which often recognize multiple cancer antigens, TIL therapy is designed to overcome some of these hurdles.
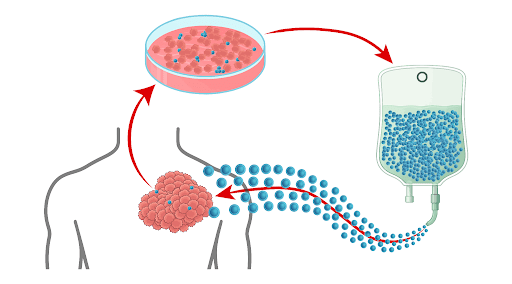
TIL Therapy: Using Immune Cells from the Tumor
In TIL therapy, a sample of a patient’s tumor is surgically removed. TILs, the T cells in the tumor, are isolated from the surgical sample, and their numbers are greatly expanded in the laboratory. With something akin to the tumor’s “zip code” in their memory, this expanded TIL population can migrate back to the tumor when infused into the patient’s bloodstream for treatment.
Patients receive chemotherapy before TIL infusion and a drug called interleukin 2 (IL-2) after the infusion. These treatments help TILs expand and function effectively inside the body. The most common side effects of TIL therapy, such as low blood counts and fevers, are primarily caused by these treatments. While these side effects can be severe, they are usually predictable and temporary.
In the pivotal clinical trial for lifileucel, the first FDA-approved cancer therapy to use TILs, this treatment was given to patients with advanced melanoma that had progressed despite standard therapy. About a third of patients had a significant decrease in the size of their tumors after lifileucel treatment. Notably, almost a third of those who responded to treatment had no additional cancer growth or spread at the end of the five-year study.
Early clinical trials suggest that TIL therapy may also hold promise for cancers beyond melanoma. Initial results have been encouraging for several other types of cancers such as lung cancer and cervical cancer.
Challenges and Limitations
Despite its potential, TIL therapy still presents significant challenges:
Patient eligibility: Patients must be healthy enough to undergo surgery, chemotherapy, and IL-2 treatment, which limits eligibility.
Production time: After a tumor sample is collected during surgery, it takes weeks to produce enough TILs for infusion, during which the cancer may progress.
Variability: Because TILs come from a patient’s own tumor, the quality and potency of the TIL product can vary from person to person.
Can alternative regimens have the same efficacy as chemotherapy and IL-2? How can we optimize the expansion and production of TILs to generate a more potent product more quickly? These are some of the questions that researchers are addressing to make TIL therapy safer and more effective.
The Future of TIL Therapy
Looking ahead, researchers are also identifying and studying biomarkers to advance therapy. Biomarkers are patient and tumor characteristics that help predict treatment outcomes or side effects. By using biomarkers, clinicians can develop more personalized therapies that give patients better outcomes with fewer side effects. In addition, selecting specific characteristics of the TIL product itself may help generate more potent therapies.
Ultimately, combining different approaches may yield the best results. In clinical trials, TIL therapy is being added to regimens with immune checkpoint inhibitors, which are now first-line therapy for a variety of cancers. In other studies, genetic engineering is being used to modify TILs to increase their potency.
Conclusion
Lifileucel was the first to show that TIL therapy can improve patient outcomes. Now, new technologies and insights into T-cell biology are paving the way to new advances. Together, these next-generation strategies aim to expand the reach and impact of TIL therapy for patients with solid tumors.
Header Image Source: Created by the author using AI-generated components (Microsoft Copilot); assembled and edited in Adobe Illustrator.
Edited by Hina Umer
References
1. Brudno JN, Maus MV, Hinrichs CS. CAR T cells and T-cell therapies for cancer: a translational science review. JAMA. 2024;332(22):1924. doi:10.1001/jama.2024.19462
2. Chen R, Johnson J, Rezazadeh A, Dudek AZ. Tumour-infiltrating lymphocyte therapy landscape: prospects and challenges. BMJ Oncol. 2025;4(1):e000566. doi:10.1136/bmjonc-2024-000566
3. Coukos G, Donia M, Gastman BR, et al. The science of tumor-infiltrating lymphocytes (TIL): perspectives from the SITC Surgery Committee. J Immunother Cancer. 2025;13(11):e013420. doi:10.1136/jitc-2025-013420
4. Medina T, Chesney JA, Kluger HM, et al. Long-term efficacy and safety of lifileucel tumor-infiltrating lymphocyte cell therapy in patients with advanced melanoma: a 5-year analysis of the C-144-01 study. J Clin Oncol. 2025;43(33):3565-3572. doi:10.1200/JCO-25-00765
5. Navarro Rodrigo B, Ortiz Miranda Y, Corria-Osorio J, Coukos G, Harari A. Immune correlates and mechanisms of TIL therapy efficacy: current insights and knowledge gaps. Trends Cancer. 2025;11(10):993-1004. doi:10.1016/j.trecan.2025.08.002

Leave a comment