Reading time: 6 minutes
Chris Wang
From the initial discovery in the early 2000s that low intensity, intermediate frequency (150-200 KHz), alternating electric fields can inhibit growth of cancer cells, the use of tumor treating fields (TTFields) has continued to expand. As of 2026, devices that deliver TTFields, made by the company Novocure, have landed FDA approvals for 4 different difficult to treat cancer types as shown in Table 1, and results from an ongoing phase 3 trial in ovarian cancer (NCT03940196), suggest an additional approval may lie in the near future.
| Year | FDA Approval | Device Name |
| 2011 | Recurrent/refractory glioblastoma (GBM) as monotherapy | Optune Gio® |
| 2015 | Newly diagnosed GBM in combination with adjuvant temozolomide after surgery and concurrent chemoradiation with temozolomide | Optune Gio® |
| 2019 | Malignant pleural mesothelioma (MPM) in combination with pemetrexed and platinum-based chemotherapy[Approved under Humanitarian Device Exemption Pathway] | Optune Lua® |
| 2024 | Metastatic non-small cell lung cancer (NSCLC) in combination with a PD-1/PD-L1 inhibitor or docetaxel after failure of platinum-based chemotherapy | Optune Lua® |
| 2026 | Locally advanced pancreatic ductal adenocarcinoma (PDAC) in combination with gemcitabine and nab-paclitazel | Optuna Pax® |
Table 1. Timeline of FDA Approvals for Currently Commercially Available TTField Devices
The mechanisms in which TTFields are thought to block tumor growth are highlighted in a previous OncoBites article listed here. In summary, TTFields are thought to disrupt microtubule assembly and intracellular components through effects on charged macromolecules. This disruption of microtubule function damages tumor cells because fast-growing cancer cells heavily rely on microtubules to form the mitotic spindle, align & separate chromosomes, and position the contractile ring during cell division. TTFields differ from external beam radiation therapy by utilizing continuous, non-ionizing levels of electromagnetic radiation to disrupt cellular processes critical for rapid cell division. Due to this property, TTFields can be safely administered for extended periods of time without interruption.
Side effects of TTFields are generally mild and limited to skin irritation, sweating, and itching beneath the electrodes. However, issues surrounding its cost and impact on the quality of life of patients remain barriers against widespread adoption. Patient-reported concerns about TTFields include the invasive requirement to wear the device for at least 18 hours/day, the need to replace device arrays two to three times per week, as well as the high visibility of therapy. For a median treatment duration of 3 to 8 months seen within trials studying TTFields, these patient-related concerns should be taken into account when deciding on treatment.
Despite the burdens of treatment, TTFields offer positive benefits as shown in the landmark studies in glioblastoma (GBM), non-small cell lung cancer (NSCLC), and pancreatic ductal adenocarcinoma (PDAC) listed in Table 2.
In the EF-14 trial, TTFields, when added to standard of care (SOC) treatment for newly diagnosed GBM, improved the median amount of time patients did not have their tumor grow by more than 2 months and the amount of time patients remained alive by almost 5 months. Because the historical median survival rate of GBM patients is a dismal 12 to 15 months, this 2 month benefit in time to tumor growth and 5 month benefit in survival stands out. Additionally, because the SOC for newly diagnosed GBM had not changed since the 2005 approval for the chemotherapy pill temozolomide, approval of TTFields in 2015 electrified the idea that GBM treatments could be improved upon. Ongoing trials studying TTfield in newly diagnosed GBM look to expand its indication into the concurrent chemoradiation phase (NCT04471844) and in combination with an immune checkpoint inhibitor and adjuvant temozolomide (NCT06556563).
Similarly, in the LUNAR trial, supplementing SOC with TTFields in pre-treated NSCLC patients, improved the median amount of time patients remained alive by around 3 months compared to SOC treatments alone. However, in this study, TTFields did not slow the time to cancer growing or lead to tumor shrinkage in a greater number of patients compared to SOC alone. Therefore, critics argue that the improvement in overall survival could have been related to a confounding factor rather than a direct benefit from TTFields. Nevertheless, the FDA approved TTFields for relapsed/refractory metastatic NSCLC because there are currently very few treatments available for patients who fail first line therapy and lack a targetable genetic abnormality.
Lastly, in the PANOVA-3 trial, TTFields, when added to chemotherapy for locally advanced PDAC, improved the median amount of time patients remained alive by 2 months compared to chemotherapy alone. With a historical 1 year survival rate of <50% for locally advanced PDAC that cannot be surgically removed, this benefit of 2 months remains notable. Similar to the LUNAR trial, TTFields did not slow disease progression or lead to tumor shrinkage in the PANOVA-3 trial. However, TTFields improved pain-free survival; an important endpoint given pancreatic cancer patients experience high rates of disease-related pain due to frequent tumor invasion into nerve-rich environments.
| Trial Name | Population | Intervention/Control | Outcomes |
| EF-14 | N=695 patients with newly diagnosed GBM after surgery and simultaneous chemoradiation | Randomized 2:1 to TTFields delivered to the scalp continuously plus SOC vs SOC alone (adjuvant temozolomide) | Tumor growth was slowed in patients receiving TTFields Median: 6.7 months vs 4.0 months Lengths of survival increased for patients receiving TTFields Median: 20.9 months vs 16.0 months |
| LUNAR | N=276 patients with relapsed/refractory NSCLC that were pre-treated with a platinum based chemotherapy regimen | Randomized 1:1 to TTFields delivered to the thoracic region continuously plus SOC vs SOC alone (investigator’s choice of immune checkpoint inhibitor or docetaxel) | Lengths of survival increased for patients receiving TTFields Median: 13.2 months vs 9.9 months [Survival benefit seen mostly in patients receiving an immune checkpoint inhibitor and not with patients receiving docetaxel] |
| PANOVA-3 | N=571 newly diagnosed locally advanced PDAC that cannot be surgically removed | Randomized 1:1 to TTFields delivered to the abdominal region continuously plus chemotherapy vs chemotherapy alone (gemcitabine + nab-paclitaxel) | Lengths of survival increased for patients receiving TTFields Median16.2 months vs 14.2 months Lengths of time without worsening cancer-related pain increased for patients receiving TTFields Median15.2 months vs 9.1 months |
Table 2. Summary of Landmark Studies on TTFields in GBM, NSCLC, and PDAC
In summary, TTFields have now grown beyond their initial approval in GBM into NSCLC and PDAC with ongoing trials aimed to identify more cancer types that respond well. Despite challenges in the delivery method, TTFields are a safe treatment that have demonstrated increased survival across multiple cancers.
Abbreviations:
FDA: Food and Drug Administration
GBM: glioblastoma
KHz: kilohertz
MPM: malignant pleural mesothelioma
NSCLC: non-small cell lung cancer
PD-1: programmed cell death protein 1
PD-L1: programmed death-ligand 1
PDAC: pancreatic ductal adenocarcinoma
SOR: standard of care
TTF: tumor treating fields
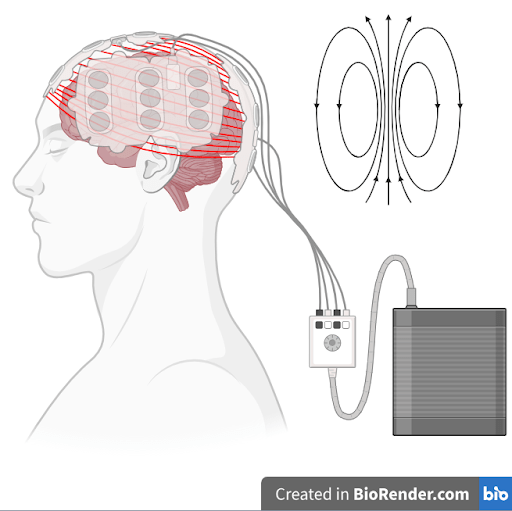
Header Image Caption and Source: Tumor treating fields for glioblastoma with arrays applied to scalp and a portable power source. Created by author in Biorender.com
Edited by Cameron Van Cleave
References
- Khagi S, Kotecha R, Gatson NTN, Jeyapalan S, Abdullah HI, Avgeropoulos NG, Batzianouli ET, Giladi M, Lustgarten L, Goldlust SA. Recent advances in Tumor Treating Fields (TTFields) therapy for glioblastoma. Oncologist. 2025 Feb 6;30(2):oyae227.
- Kirson ED, et al. Alternating electric fields arrest cell proliferation in animal tumor models and human brain tumors. Proc Natl Acad Sci U S A. 2007 Jun 12;104(24):10152-7.
- Stupp R, et al. NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer. 2012 Sep;48(14):2192-202.
- Stupp R, et al. Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma: A Randomized Clinical Trial. JAMA. 2017 Dec 19;318(23):2306-2316.
- Ceresoli GL, et al. Tumour Treating Fields in combination with pemetrexed and cisplatin or carboplatin as first-line treatment for unresectable malignant pleural mesothelioma (STELLAR): a multicentre, single-arm phase 2 trial. Lancet Oncol. 2019 Dec;20(12):1702-1709.
- Leal T, et al. Tumor Treating Fields therapy with standard systemic therapy versus standard systemic therapy alone in metastatic non-small-cell lung cancer following progression on or after platinum-based therapy (LUNAR): a randomised, open-label, pivotal phase 3 study. Lancet Oncol. 2023 Sep;24(9):1002-1017.
- Babiker HM, et al. Tumor Treating Fields With Gemcitabine and Nab-Paclitaxel for Locally Advanced Pancreatic Adenocarcinoma: Randomized, Open-Label, Pivotal Phase III PANOVA-3 Study. J Clin Oncol. 2025;43(21):2350-2360.

Leave a comment