Reading time: 3 minutes
Tala Tayoun
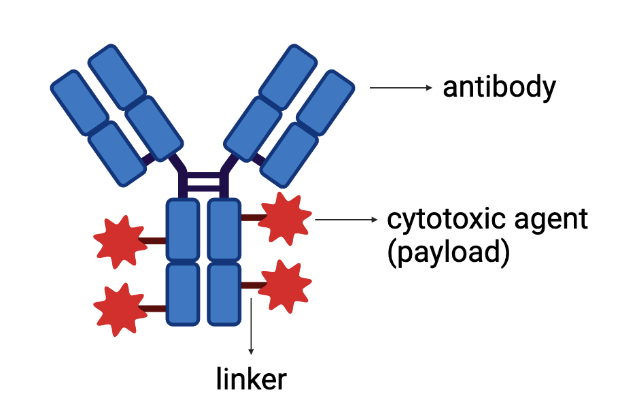
Antitumor antibody-drug conjugates (ADCs) are a class of therapeutic molecules used in cancer therapy. Their main purpose is to channel the drug to a specific tumor site, kill cancer cells while sparing healthy cells and minimizing collateral damage [1]. ADC comprises of three key components: (1) an antibody, (2) a chemical linker and (3) a cytotoxic payload (drug) (Figure 1):
Figure 1. Antibody-drug conjugate complex structure. Created using Biorender.
1. The antibody component is designed to recognize a well-characterized molecule homogeneously expressed on the surface of the tumor cell, i.e. the antigen. The ideal antigen must be extracellular to allow the ADC-target complex to be subsequently internalized via endocytosis (see below). Ideally, the epitope would present very low expression on healthy cells, which allows the ADC to selectively target tumor cells while avoiding healthy cells.
2. The linker connects the antibody to the cytotoxic agent. It is stable in the blood and makes sure that the antibody is directed towards a specific cancer cell surface molecule and remains intact until it reaches it. The linker is then able to release the drug payload inside tumor cells.
3. The cytotoxic payload is the drug which will be responsible for killing the cancer cell upon release from the linker.
When an ADC is administered intravenously to a patient, it circulates throughout the bloodstream until it attaches specific tumor cells that express the target antigen. The ADC is then internalized into the cancer cell interior. Inside the cell, the chemical linker is cleaved or degraded, thus releasing the cytotoxic drug, which then exerts its toxic effects and kills specifically the tumor cell. Some of the payload categories include DNA-damaging agents or tubulin inhibitors.
The last decade has been a real game changer in the field of cancer targeted therapy using ADCs. Recent technological advances in bioengineering to improve ADC design have drastically accelerated their clinical development. There are currently 13 FDA-approved ADCs on the market worldwide, in both hematological (e.g. leukemia, lymphoma) and solid-tumor malignancies (e.g. breast, ovarian cancers) [2].
There are currently hundreds of ADC agents being tested in clinical trials [2]. Among others, exciting findings from the phase III MIRASOL study (Clinical trial information: NCT04209855) were recently reported in ovarian cancer [3]. The clinical trial showed significantly improved survival benefit in patients treated with Mirvetuximab soravtansine (MIRV), an ADC targeting a specific type of ovarian cancer (folate receptor alpha-positive), compared to conventional chemotherapy.
The field of ADC development and research is evolving rapidly and major efforts are being made to improve the efficacy and safety of these therapies.
Edited by Pratyusha Ghanta
References:

Leave a comment