Reading time: 4 minutes
Megan Majocha
Can you imagine trying to put together clear, acrylic puzzle pieces? It is difficult to put together all the unknowns without a picture. Metastasis research is similar – trying to put the pieces together without knowing exactly how metastatic cells function in the human body. While cancer metastasis research is advancing and we have a better understanding of how metastasis works, it remains a challenge.
There are several reasons why metastasis research is complicated. For example, not all proteins in our cells are well-characterized, meaning there are not sufficient published papers about these proteins. This means researchers will need to study its function in depth without a clear picture of what the protein does, then test its function in metastasis. This is one of the reasons why published papers are critical. Researchers can eliminate and narrow down all potential mechanisms of the protein, and focus on the mechanism of interest to further explore protein function.
There are a few other reasons why metastasis research can be complicated, and may slow down research progress. First, there are different forms of one gene, known as isoforms, which can arise from alternative splicing. Most researchers focus on one isoform of a gene, which may have a different function or localization in the cell than the other isoform of the same gene. Furthermore, each isoform may bind to different proteins. Because of these special differences, it takes a lot of effort to characterize every known isoform of the same gene, which requires a lot of resources and can be expensive.
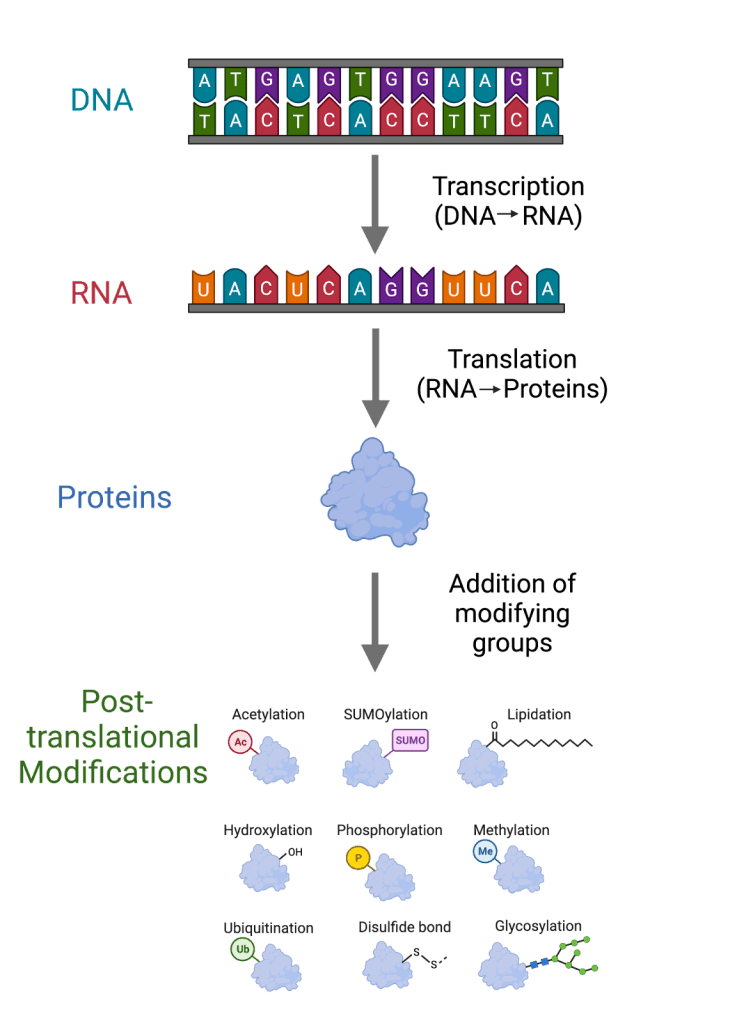
Another challenge is post-translational modifications (PTMs), which are modifications that can occur on proteins after they are translated from mRNA and are critical for regulating protein function, localization, stability, and interactions. The information contained in DNA is replicated (DNA replication), the DNA information is copied into mRNA (transcription), and the mRNA information is then translated into proteins (translation) (Figure 1). PTMs are involved in many cellular processes, including metastasis. What do PTMs have to do with the complexity of metastasis research, you ask? Many proteins involved in the complex metastatic process undergo PTMs and can either promote or inhibit metastasis. The effects of PTMs on protein function can vary depending on the cellular environment. Thus, the complexity of PTMs makes it challenging to study their effects on metastasis. Understanding the detailed and comprehensive role of PTMs in metastasis is critical, and will help develop new treatments and interventions for patients.
A further difficulty is the use of cell lines, which are a common tool in biomedical research. They are easy to use, cost-effective, and widely available. Cell lines are commonly used in screening assays to test the efficacy of potential cancer treatments. However, there are limitations. Cell lines are often derived from a single tumor or metastatic site. Cancer is a highly heterogeneous disease, meaning the tumor and its metastases have different genetic and molecular profiles. Additionally, cell lines are adapted to cell growth in culture over generations, meaning that they are grown on plastic in a flask without its normal microenvironment, which consists of other cell types and tissues surrounding cancer cells, which can affect cancer and metastasis progression. This adaptation can lead to genetic and phenotypic changes and may not reflect cancer cells in the human body. To overcome some of these cell line issues, researchers are currently trying to incorporate organoids in their studies. Organoids are cells that are grown in 3D culture rather than 2D and mimic key biological complexity of the organ, as well as its function and structure.
It is critical to understand the molecular mechanisms of metastasis to develop better therapies. While metastasis research is advancing, there are multiple limitations, some of which are outlined here. Metastasis is a complex biological process that consists of multiple genetic and molecular changes, throughout the body, making metastasis research challenging. Publishing negative results is helpful for other researchers, so they can avoid duplicating their efforts and can help them refine their hypotheses. This will save researchers a lot of time. Unfortunately, the publication of negative results does not often happen because, in general, there is a bias toward publishing positive results as they are seen as more exciting than negative results1.
Edited by Ana Isabel Castillo Orozco
Reference:
- Mlinarić, A., Horvat, M., & Šupak Smolčić, V. (2017). Dealing with the positive publication bias: Why you should really publish your negative results. Biochemia medica, 27(3), 030201. https://doi.org/10.11613/BM.2017.030201


Leave a comment