Reading time: 5 minutes
Patty Spears
The applause was thunderous and traveled like a wave across the large auditorium at the end of a 2022 American Society of Clinical Oncology (ASCO) Annual Meeting Plenary Session talk. It did not stop. The audience stood and kept applauding and reveling in the positive results of a clinical trial – DESTINY Breast04. This moment was something I will never forget.
What led to this clinical trial, and why did this study lead to such an enthusiastic response?
It is a combination of a few things:
- The type of drug
- More patients may benefit from this type of drug
- A new category of an old biomarker
The type of drug: Antibody Drug Conjugates (ADCs)
The development of new treatments for cancer has taken many routes. One new way to treat cancer is to use a new drug that contains an antibody and chemotherapy together, or antibody-drug conjugate (ADC).
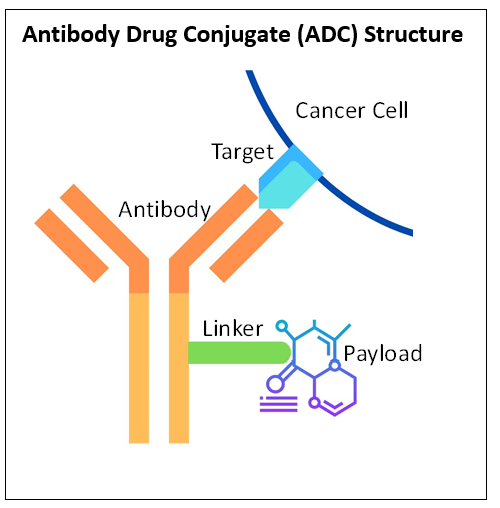
What are antibody-drug conjugates (ADCs)
There are three parts to ADCs: an antibody, a linker, and a payload. They all have different functions. (See the box Antibody Drug Conjugate Structure for more details)
- The antibody finds and binds to the cancer cell
- The linker attaches the antibody to the payload
- The payload is often a chemotherapy drug known to treat cancer
How do ADCs work?
Many have described ADCs as Trojan horses. Typically, cancer is treated with chemotherapy. Yet, chemotherapy tends to kill both cancer cells and other healthy cells. This means patients treated with chemotherapy often suffer many unwanted toxicities and side effects. ADCs link chemotherapy to an antibody in a way to make the chemotherapy not active until delivered to the cancer cell by the antibody. The antibody can tell the difference between cancer cells and healthy cells, and once it finds the cancer, it can attach to it. Then the linker releases the chemotherapy in and around the cancer cell.
The development of ADCs to treat cancer
There have been many advances in the optimization of this type of cancer treatment over the past 30 years. A review by Tarantino et. al. (1) describes the timeline of the development of ADCs for cancer treatment. ADC development began in 1990 with their first clinical use in solid cancers. As of 2021 there have been 7 ADC approvals for use in different cancers. ADCs are now on the frontlines of treatment strategy for solid tumor cancers.

Patients who may benefit: ADCs in Breast Cancer
In 2013, trastuzumab emtanzine (T-DM1) was the first ADC approved to treat metastatic HER2 positive breast cancer. In 2019, its approval expanded to include early stage HER2 positive breast cancer. The 2013 approval spurred the development of more ADCs with different and improved characteristics. One of the new ADCs is trastuzumab deruxtecan (T-DXd). T-DXd uses the same targeting antibody (trastuzumab), but uses a different linker (now cleavable) and a different drug payload. T-DXd also has more drug linked to each antibody to increase cancer cell killing. In 2019, T-DXd was approved to treat HER2 positive metastatic breast cancer. Another ADC, sacituzumab govitecan, had several differences from the previous ADCs. Sacituzumab govitecan has a different antibody, linker and payload. The antibody (sacituzumab) targets a different molecule found in many breast cancers called TROP-2. It also has a better linker and a different payload that can pass through cell membranes. There is also more drugs linked to each antibody. In 2020, this ADC was approved to treat triple negative metastatic breast cancer in patients who have already undergone many treatments and who need new options. This was an important approval for these patients. (See the box ADC Clinical Trials for key studies leading to approval of ADCs in metastatic breast cancer.)
New category of an old biomarker: Recent exciting news in breast cancer treatment with T-DXd
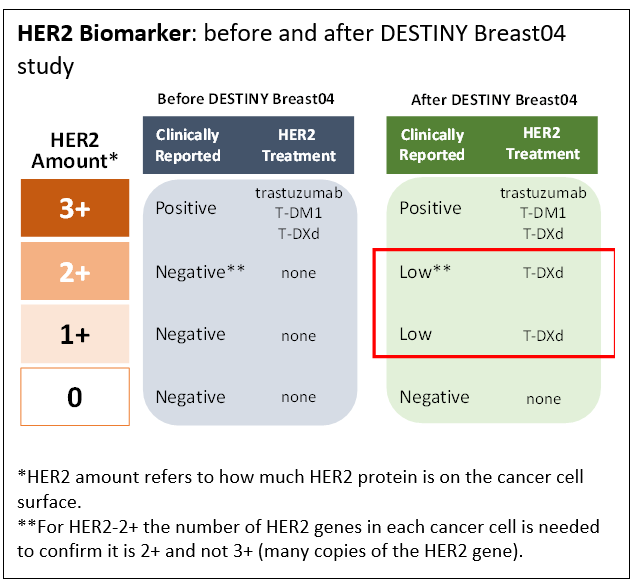
Results presented from the recent T-DXd study, DESTINY-Breast04 (2) made a big splash at the recent 2022 ASCO Annual meeting, prompting the unforgettable standing ovation. The antibody used in T-DXd, trastuzumab, is a well-known treatment for patients with HER2 positive breast cancer. However, the DESTINY-Breast04 study looked at how well T-DXd worked in patients with HER2-Low breast cancer tumors. They found that T-DXd worked in patients with low HER2 expressing breast cancer. (See the box HER2 Biomarker)
Why did this result deserve a standing ovation?
- HER2-Low is a new biomarker for a targeted treatment for metastatic breast cancer.
- Patients with triple negative breast cancer that is HER2-Low have a new, targeted treatment option.
- More patients may benefit from receiving T-DXd.
There is still a lot to consider going forward.
- There are some toxicities that need to be watched and controlled. This is particularly important when considering treating patients with early stage cancer. Early stage breast cancer is more likely to be survivable, so toxicities should be minimized.
- It is not clear when, or in what order, to add these new treatments to current treatments.
- Diagnostic HER2 testing needs to include a new HER2-Low category.
This study is already making a positive impact in the clinic. Many patients who are being treated for triple negative breast cancer are asking if their cancer is actually HER2-Low. If it is, there is now a new, targeted drug for them to use to treat their cancer. Having a new, targeted treatment option is especially meaningful for patients facing the end of chemotherapy options.
On August 5, 2022 “… the U.S. Food and Drug Administration approved Enhertu (fam-trastuzumab-deruxtecan-nxki), an IV infusion for the treatment of patients with unresectable (unable to be removed) or metastatic (spread to other parts of the body) HER2-low breast cancer. This is the first approved therapy targeted to patients with the HER2-low breast cancer subtype, which is a newly defined subset of HER2-negative breast cancer.” (3)

Edited by Gabrielle Dardis
References:
- Tarantino P, Carmagnani Pestana R, Corti C, Modi S, Bardia A, Tolaney SM, Cortes J, Soria JC, Curigliano G. Antibody-drug conjugates: Smart chemotherapy delivery across tumor histologies. CA Cancer J Clin. 2022 Mar;72(2):165-182. doi: 10.3322/caac.21705. Epub 2021 Nov 12. PMID: 34767258.
- Modi S, Jacot W, Yamashita T, Sohn J, Vidal M, Tokunaga E, Tsurutani J, Ueno NT, Prat A, Chae YS, Lee KS, Niikura N, Park YH, Xu B, Wang X, Gil-Gil M, Li W, Pierga JY, Im SA, Moore HCF, Rugo HS, Yerushalmi R, Zagouri F, Gombos A, Kim SB, Liu Q, Luo T, Saura C, Schmid P, Sun T, Gambhire D, Yung L, Wang Y, Singh J, Vitazka P, Meinhardt G, Harbeck N, Cameron DA; DESTINY-Breast04 Trial Investigators. Trastuzumab Deruxtecan in Previously Treated HER2-Low Advanced Breast Cancer. N Engl J Med. 2022 Jul 7;387(1):9-20. doi: 10.1056/NEJMoa2203690. Epub 2022 Jun 5. PMID: 35665782.
- FDA approves first targeted therapy for HER2-low breast cancer. News release. FDA. August 5, 2022. Accessed August 5, 2022. https://bit.ly/3d4X2JQ
- ASCO Daily News, https://dailynews.ascopubs.org/do/10.1200/ADN.22.201047/full/
FDA Approval References:
Kadcyla® (T-DM1) ado-trastuzumab emtansine; Genentech, Inc.
Amiri-Kordestani L, Blumenthal GM, Xu QC, Zhang L, Tang SW, Ha L, Weinberg WC, Chi B, Candau-Chacon R, Hughes P, Russell AM, Miksinski SP, Chen XH, McGuinn WD, Palmby T, Schrieber SJ, Liu Q, Wang J, Song P, Mehrotra N, Skarupa L, Clouse K, Al-Hakim A, Sridhara R, Ibrahim A, Justice R, Pazdur R, Cortazar P. FDA approval: ado-trastuzumab emtansine for the treatment of patients with HER2-positive metastatic breast cancer. Clin Cancer Res. 2014 Sep 1;20(17):4436-41. doi: 10.1158/1078-0432.CCR-14-0012. Epub 2014 May 30. PMID: 24879797.
ENHERTU® (T-DXd) fam-trastuzumab deruxtecan; Daiichi Sankyo
Narayan P, Osgood CL, Singh H, Chiu HJ, Ricks TK, Chiu Yuen Chow E, Qiu J, Song P, Yu J, Namuswe F, Guiterrez-Lugo M, Hou S, Pierce WF, Goldberg KB, Tang S, Amiri-Kordestani L, Theoret MR, Pazdur R, Beaver JA. FDA Approval Summary: Fam-Trastuzumab Deruxtecan-Nxki for the Treatment of Unresectable or Metastatic HER2-Positive Breast Cancer. Clin Cancer Res. 2021 Aug 15;27(16):4478-4485. doi: 10.1158/1078-0432.CCR-20-4557. Epub 2021 Mar 22. PMID: 33753456; PMCID: PMC8570919.
TRODELVY® (Saci) sacituzumab govitecan; Immunomedics, Inc.
Wahby S, Fashoyin-Aje L, Osgood CL, Cheng J, Fiero MH, Zhang L, Tang S, Hamed SS, Song P, Charlab R, Dorff SE, Ricks TK, Barnett-Ringgold K, Dinin J, Goldberg KB, Theoret MR, Pazdur R, Amiri-Kordestani L, Beaver JA. FDA Approval Summary: Accelerated Approval of Sacituzumab Govitecan-hziy for Third-line Treatment of Metastatic Triple-negative Breast Cancer. Clin Cancer Res. 2021 Apr 1;27(7):1850-1854. doi: 10.1158/1078-0432.CCR-20-3119. Epub 2020 Nov 9. PMID: 33168656.


Leave a comment