Reading time: 4 minutes
Kate Secombe
You may have read previously on OncoBites about immunotherapies, and the great promise they hold in treating a wide range of cancers. Commonly used immunotherapies include checkpoint inhibitors such as PD-1 or CTLA-4 inhibitors. Here I will be looking at an emerging subset of immunotherapies known as immune agonist antibodies (IAAs). There is a wide range of active and upcoming early-stage clinical trials currently investigating IAAs, in combination with other immunotherapies.
So, what is an immune agonist antibody? An agonist is a molecule or a compound that can activate a cellular receptor to induce a biological response. IAAs, therefore, activate particular receptors on immune cells to generate a specific immune response against cancer cells via induction of processes like the increased proliferation of immune cells, tumor cell-killing activity, and immune cell survival. While there is a variety of IAAs in development, targeting a range of receptors, the CD40 and CD137 IAAs will be focussed on here.
What do immune agonist antibodies target?
The function of immune cells in our body is tightly regulated by co-stimulatory receptors. Co-stimulation is a second signal, that in the presence of an antigen-presenting cell, is often required for an immune cell to mount an immune response. There is genetic evidence for the role of co-stimulatory molecules in mediating anti-cancer immune responses.
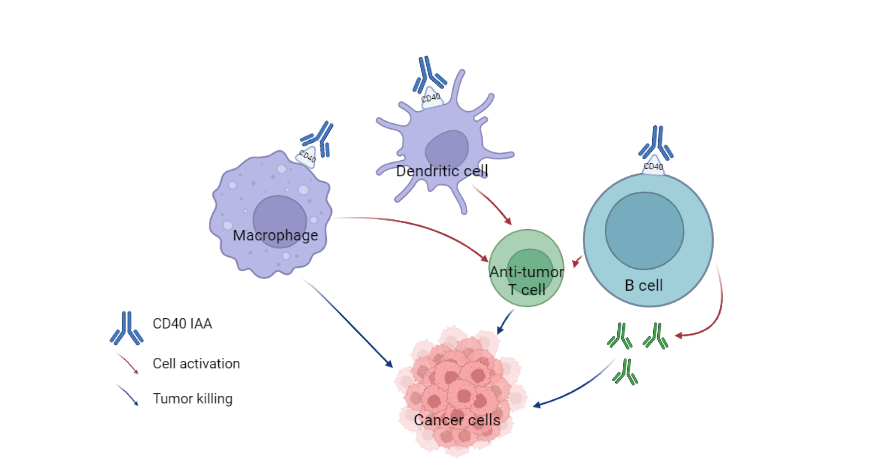
CD40 and CD137 are two of these co-stimulatory molecules. CD40 and CD137 IAAs have been developed to specifically bind to the CD40 or CD137 molecules, respectively, generating an immune response. Developing these agonist antibodies can be a difficult process, often requiring the trialing of many potential candidates in a range of pre-clinical experiments. The most important feature of IAAs is that they bind and activate their target receptor in such a way that closely mimics what would normally happen in the human body.
Upon activation, CD40 can cause dendritic cells to activate antitumor, or cytotoxic T cells, ensuring that macrophages destroy structural cells in the tumor (see Figure 1). CD137 is expressed in immune cells such as dendritic cells, natural killer cells, and T cells. Anti-CD137 antibodies have also been shown to be able to activate cytotoxic T cells specific to tumor cells.
How is this a useful cancer treatment?
CD40 is expressed mainly on antigen-presenting cells (like dendritic cells). CD40 IAAs currently in development, therefore, work predominantly by inducing antigen presentation, which leads to the immune response, directed towards tumor cells, described above. Unfortunately, trials of CD40 IAA treatment alone have not shown excellent results so far, so the current focus is on combination therapy with other immunotherapies.
CD137 IAAs can stimulate and activate effector T cells, natural killer cells, and macrophages to kill tumor cells. Interestingly, CD137 IAAs can increase the amount of antigen-specific memory T cells as well, which can result in a longer-lasting immune response against cancer cells. Further research in this area is also looking at combination therapies, as well as applications for CD137 IAAs in different cancer stages, and tumor types.
Side effects of IAA treatment
Unfortunately, many studies of IAAs have shown dose-limiting toxicities. These side effects have typically been immune-mediated and may include cases of cytokine release syndrome, which manifests in the minutes to hours after drug administration as fever, chills, and sometimes headache and pain. In addition, CD40 administration has also been associated with liver function abnormalities.
Current research has suggested that the gut microbiome may be involved in the mediation of these toxicities, and other studies have examined whether we could avoid these toxicities by administering IAA locally (i.e. directly to the tumor instead of systemically via blood or oral medication). In addition, researchers have looked at whether co-administering immunosuppressive steroid medications at the same time may reduce these side effects.
Next steps
Various IAAs have been tested clinically for more than a decade now, however, none have yet been approved for widespread clinical use, showing the difficulty in developing specific IAAs that work with minimal side effects. There is still a lot of work to do to fully understand how best to use IAAs in combination with other therapies or alone, in addition to minimizing side effects. Further complicating this dilemma is that previous clinical trials have shown that some IAA therapies are highly effective in one patient, but not in another, even with the same cancer diagnosis.
Edited by Maha Said
Major works referenced
Mayes PA, Hance KW, Hoos A. The promise and challenges of immune agonist antibody development in cancer. Nat Rev Drug Discov. 2018 Jul;17(7):509-527. doi: 10.1038/nrd.2018.75.
Vonderheide RH. CD40 Agonist Antibodies in Cancer Immunotherapy. Annu Rev Med. 2020 Jan 27;71:47-58. doi: 10.1146/annurev-med-062518-045435.
Chu DT, Bac ND, Nguyen KH, Tien NLB, Thanh VV, Nga VT, Ngoc VTN, Anh Dao DT, Hoan LN, Hung NP, Trung Thu NT, Pham VH, Vu LN, Pham TAV, Thimiri Govinda Raj DB. An Update on Anti-CD137 Antibodies in Immunotherapies for Cancer. Int J Mol Sci. 2019 Apr 12;20(8):1822. doi: 10.3390/ijms20081822.