Reading time: 5 minutes
Aishat Motolani
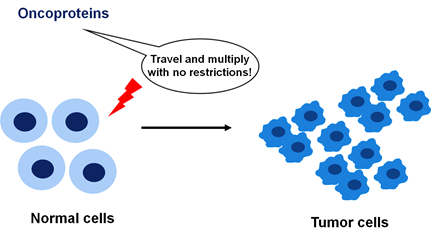
Just as bulldozers work to demolish dilapidated buildings or clear weeds on farmlands, PROteolysis TArgeting Chimeras (PROTACs) are small bifunctional molecules designed to degrade proteins that are present in excess in diseased conditions. In cancer, these specific proteins are called oncoproteins. Oncoproteins are capable of transforming a normal cell into a cancerous one, leading to sprawling tumor growth (Figure 1). Since the initial proof of concept for PROTAC two decades ago, several pharmaceutical companies have successfully developed PROTACs to target major proteins that contribute to a disease progression. But before we dive into the mechanism of PROTAC and its use in cancer drugs, let us briefly review the normal cellular process of protein bulldozing.
Cells maintain homeostasis partly by balancing the production and elimination of several molecules. Particularly, proteins’ abundance is tightly regulated by a cell disposal machinery called the proteasome. Proteasomes are complexes that mainly comprise a catalytic core and a regulatory subunit. As shown in Figure 2, they break down proteins that are attached to a small molecule called ubiquitin (Ub). For a protein to be degraded through the ubiquitin-proteasome system (UPS), an activating enzyme (E1) activates ubiquitin and then transfers it to a conjugating enzyme, E2. After this step, together with the ligating enzyme (E3), ubiquitin is transferred from E2 to the protein of interest.
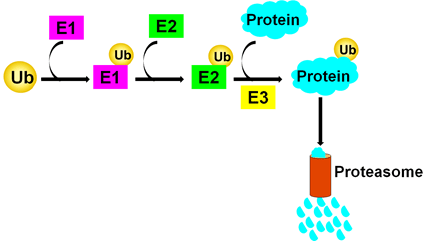
Once a protein is tagged with ubiquitin, it becomes destined for proteasomal processing or degradation. In the case of degradation, the regulatory subunit binds to the ubiquitin-tagged protein, opens the gate to the catalytic chamber, and delivers the protein into this chamber. Therein, the protein is degraded into peptides. And poof! It is gone. To this extent, old or excess proteins can be continually degraded, ensuring the abundance of functional proteins in a physiological range. In cancer, the bulldozing of oncoproteins by proteasomes is significantly slowed down or prevented.
Various oncoproteins— such as c-Myc, KRAS, NF-𝜅B, and other proteins with tongue-twisting acronyms — work tirelessly to fuel the growth, migration, and invasion of tumors into nearby tissue. In this way, the cancer cells can survive and progress in their pathologies. One strategy used by cancer cells to maintain the function of oncoproteins is to increase their stability. This can be done either through the attachment of protective groups that prevent protein degradation or through the decreased activity of the ubiquitin-proteasome system enzymes, as discussed earlier. While using proteasome activators may be appealing to trigger the degradation of oncoproteins, the resulting side effects can be dismal and widespread. Hence, PROTACs present a viable approach for reducing oncoprotein abundance in cancers.
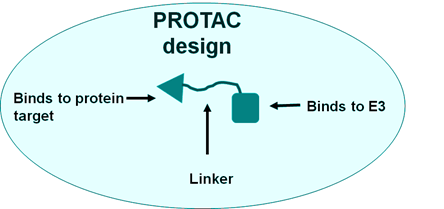
PROTAC works by targeting the degradation of a specific protein. It consists of three distinct parts: a part that binds to the protein target, another that binds to E3, and a linker that connects the protein of interest to E3 (Figure 3). This unique design of PROTAC ensures high selectivity for the degradation of specific protein targets. Some advantages provided by PROTACs in cancer therapeutics include overcoming cancer drug resistance, degrading protein targets considered undruggable, and inhibiting both catalytic and non-catalytic functions of proteins.
For example, the development of inhibitors against STAT3— a protein that induces genes that drive cancer progression— has had limited success. This is due to the lack of clear druggable sites on the oncoprotein. Interestingly, an exciting finding published in 2019 showed that a PROTAC molecule that targets STAT3, SD-36, caused complete tumor regression of leukemia and lymphoma cells in mice with no alarming toxicities.
PROTACs, like other drugs, have certain drawbacks. One of the main challenges of PROTAC-based drugs is their poor absorption into the body. This is mainly due to their bulky nature and low solubility in water. Several questions regarding PROTAC’s off-target effects, regulation of its activity, and ability to degrade extracellular proteins also require further study.
Nonetheless, PROTAC-based drugs have demonstrated remarkable potential in decreasing the growth and spread of certain cancers. In 2019, the FDA approved the first oral PROTAC drug, ARV-110, for the treatment of metastatic prostate cancer in a phase 1/2 clinical trial. This drug targets the degradation of the androgen receptor, a well-known oncoprotein that drives prostate cancer.
PROTAC has been dubbed a “game-changing technology” that is non-conforming to classical drug pharmacology. So, with more studies of the ubiquitin-proteasome systems and PROTAC-based drugs, overcoming the limitations of targeting major undruggable oncoproteins in cancer could soon be on the horizon.
Works discussed
Neklesa, T. K., Winkler, J. D., & Crews, C. M. (2017). Targeted protein degradation by PROTACs. Pharmacology & Therapeutics, 174, 138–144. doi:10.1016/j.pharmthera.2017.02.027
Gao, H., Sun, X., & Rao, Y. (2020). PROTAC technology: opportunities and challenges. ACS medicinal chemistry letters, 11(3), 237-240.
Thibaudeau, T. A., & Smith, D. M. (2019). A practical review of proteasome pharmacology. Pharmacological Reviews, 71(2), 170-197.
Bai, L., Zhou, H., Xu, R., Zhao, Y., Chinnaswamy, K., McEachern, D., Chen, J., Yang, C. Y., Liu, Z., Wang, M., Liu, L., Jiang, H., Wen, B., Kumar, P., Meagher, J. L., Sun, D., Stuckey, J. A., & Wang, S. (2019). A Potent and Selective Small-Molecule Degrader of STAT3 Achieves Complete Tumor Regression In Vivo. Cancer cell, 36(5), 498–511.e17. https://doi.org/10.1016/j.ccell.2019.10.002
Konstantinidou, M., Li, J., Zhang, B., Wang, Z., Shaabani, S., Ter Brake, F., Essa, K., & Dömling, A. (2019). PROTACs- a game-changing technology. Expert opinion on drug discovery, 14(12), 1255–1268. https://doi.org/10.1080/17460441.2019.1659242.
Header Image adapted from OpenClipart-Vectors from Pixabay

Leave a comment