Estimated reading time: 5 minutes
Payal Yokota
One of the main differences between a rapidly dividing cancer cell and a normal cell is that the cancer cells rely on glycolysis followed by lactic acid fermentation, rather than oxidative phosphorylation. This effect is called the Warburg effect, one we have previously discussed in the context of targeting cancer cell metabolism as a potential therapeutic strategy as well as specifically reviewed in the benefits of methotrexate. One way to exploit the fact that tumor cells are decorated with sugars (carbohydrates) is by using them in subunit vaccines. Subunit vaccines offer better precision in delivery and targeting cancer cells compared to the traditional whole-pathogen vaccines. Unlike live attenuated vaccines (that contain the entirety of the pathogen), subunit vaccines use only a small part of the pathogen or protein in order to provoke a response from the immune system offering a number of advantages. First, it is able to precisely seek out only those cancer cells with high sugar enrichment and synthesis (glycolysis) and therefore, fewer side-effects by virtue of avoiding normal cells. Second, this strategy can be generally applied to target a variety of cancers that have similar patterns of sugar signatures. Finally, they are relatively easy to manufacture and upscale for distribution compared to more personalized strategies. A downside to this approach is that the carbohydrate conjugate vaccines are generally regarded as less potent and require help from other additives (adjuvants) in order to enhance the immune response.
Conventionally, the lack of immunogenicity of carbohydrate (or sugar)-based vaccines has been overcome by use of adjuvants, which are substances that act to prolong or enhance antigen-specific immune response as well as controlling both the timing and location of where the antigen is being delivered. To date, however, there have only been a handful of FDA-approved adjuvants for use in vaccine design strategies. Since 1930’s, a mixture of aluminum salts (called alum) has been exclusively used in human studies. Alum works by trapping the antigen in place for efficient delivery at the precise location and time with less reported toxicity. However, one of the limitations in using alums has been their weak potency to generate a cytotoxic cellular response (Killer T cells). Additionally, the exact mechanism of how an alum traps the antigen for slow release to disease site within the body remains enigmatic. While a variety of modifications have been suggested to combine alums with higher potency adjuvant combinations, the field has hit a wall with regards to finding cheaper, faster and more efficient adjuvant targets.
In recent years, scientists have resorted to plant-based adjuvants in designing complex vaccine+adjuvant cocktails to target the enriched sugar synthesis on a variety of solid cancers such as those occurring in lungs, breasts, ovaries, skin as well as brain. Quillaja Saponaria (QS-21), an extract from the bark of Chilean trees, is a plant-based adjuvant that has been used for over 20 years. To this date, QS-21 adjuvants have only been approved for use of vaccines against malaria and shingles, but not cancer. An underlying reason why plant-based adjuvants have not been widely used or FDA-approved, is the difficulty in obtaining a sustainable supply both with regards to total yields (amounts) as well as quality. In other words, one cannot always expect to get the same concentration of the saponins from the bark of the tree, and therefore the purification process can be tedious and expensive for unreliable yields from batch to batch. Moreover, it has also been difficult to assess the exact mechanism employed by these adjuvants in order to provide precise release to the site of infection or disease, and subsequently coerce the host immune system to produce antibodies (humoral immunity) and/or activate cancer specific killer T cells (cellular immunity). While there is limited structural and mechanistic data to explain how adjuvants enhance antigen efficacy, a recent study by Pifferi et al has provided profound insights on how QS-21 adjuvants may be delivered alongside with a protein antigen, and be presented to activate cytotoxic T cells (Figure-1) suggesting a unique and critical role of adjuvants in vaccine strategies to combat cancer metastases. After being activated, these CD8+ T cells will target the tumor cells to control cancer from growing and spreading.
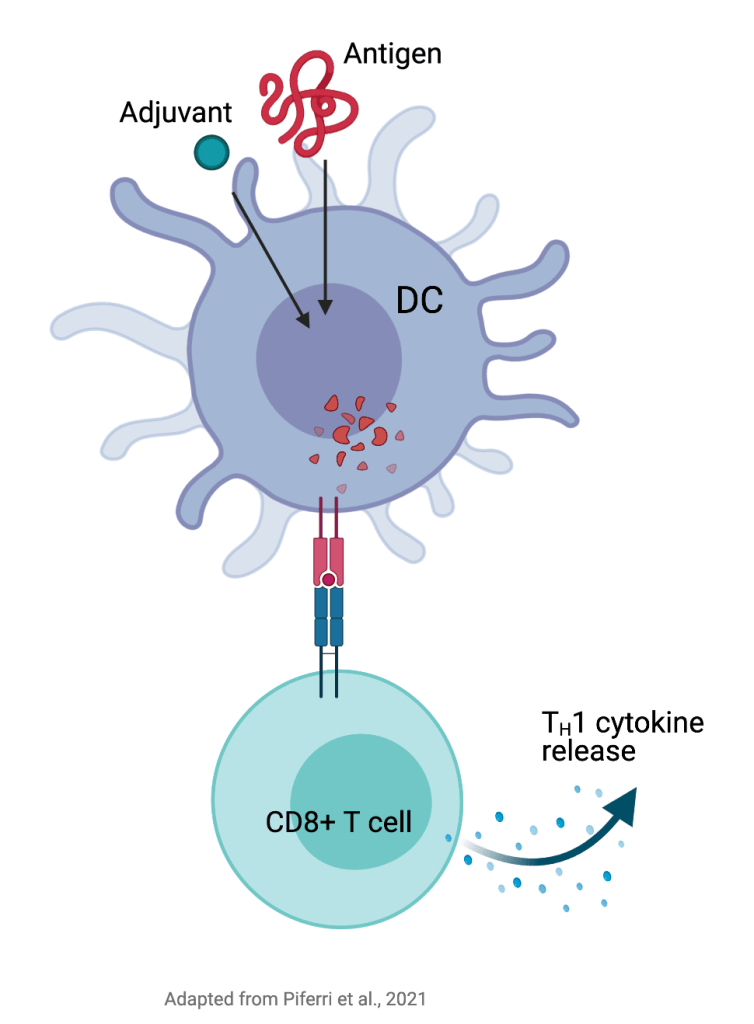
To bypass this obstacle, organic chemists have started to synthesize these plant-based adjuvants by finding chemically simpler starting points from the natural plant origins, followed by syntheses to reach the final complex structures. This strategy is most efficient because it allows for a variety of advantages. First and foremost, complex final adjuvant formulations can be designed fast and cheap, resulting in wider and sustained use. Second, by having complete control on the synthesis of the final structure, it is possible to remove the parts that induce high toxicity to the normal cells (that which causes side effects) and thereby keeping the required parts intact. Finally, these structures can be modified from initial stages to cater to a variety of cancers. While the mechanism of how these adjuvants work is not entirely elucidated, recent preclinical studies have shown convincing evidence of efficacy. What has been characterized thus far, is that upon administration by a subcutaneous injection, these complex vaccine + adjuvant cocktails reach the innate immune cells in the body that can simplify the complex structures by chopping them up into smaller pieces by processing them through molecular shredders within the cells (proteasomal machinery). Then, these tiny pieces are brought back up on the surface of the cell and presented to killer T cells, to activate them and to initiate immune protective mechanisms to fight cancer cells.
In summary, semi-synthetic plant based adjuvants can prove to be game-changers in the design and delivery of subunit vaccines that exploit the carbohydrate chemistry of a variety of cancers without the immense toxic side-effects and can be engineered safely, rapidly and economically.
Edited by Garima Khanna
Further Reading:
Pifferi, C., Fuentes, R. & Fernández-Tejada, A. Natural and synthetic carbohydrate-based vaccine adjuvants and their mechanisms of action. Nat Rev Chem (2021). https://doi.org/10.1038/s41570-020-00244-3
Petrovsky, N. & Cooper, P. D. Carbohydrate-based immune adjuvants. Expert Rev Vaccines (2012). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3118391/
Chea et al. Synthesis and preclinical evaluation of QS-21 variants leading to simplified vaccine adjuvants and mechanistic probes. J Am Chem Soc. (2013). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3118391/
https://www.vaccines.gov/basics/types
Image made using BioRender(™)

Leave a comment