Reading time: 3 minutes
Garima Khanna
Vanadium is a hard, silvery-grey, malleable transition metal. Transition metals are chemical elements that have valence electrons capable of participating in formation of chemical bonds. While the term transition has no particular chemical significance, it is a convenient name by which to distinguish the similarity of the atomic structures and resulting properties of the elements so designated.
The elemental metal is rarely found in nature and usually isolated artificially. Vanadium derived complexes have caught the interest of many scientists to treat human diseases, especially cancer and diabetes. They have shown promising effects when studied in vitro and in animals. Nevertheless, strong reluctance to explore their use prevails due to their plausible oxidative and genotoxicity associated with higher dosing of vanadium. There are also concerns over accumulation of vanadium in the long-term leading to adverse off-target tissue actions. But when considering cancer patients, a short term treatment may weigh more pressing, which has encouraged the investigation of better vanadium complexes. Upcoming technologies utilize precision medicine by targeting drugs specifically to tumors and at the same time, minimizing off target issues. A similar approach can be utilized for oxidovanadium complexes to deliver toxic ligands to tumors. The aforementioned complexes rapidly exchange ligands in the gut and release ligands which generate vanadium complexed with proteins, namely albumin and transferrin. This pre-delivery reaction offers hindrance in clinical use for cancer therapy. With this background, it would prove to be of great advantage if we can develop stable complexes and tumor specific delivery packages.
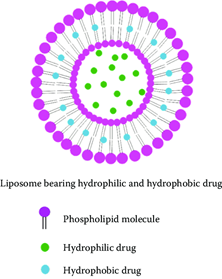 Liposomes are small artificial vesicles of spherical shape that can be created from cholesterol and natural non-toxic phospholipids. Due to their size and hydrophobic and hydrophilic character, liposomes are promising systems for drug delivery. These complexes are highly hydrophobic and are able to affect either cytotoxicity or differentiation in several neuroblastoma tumor-derived cell lines. These liposomal particles are more effective than the earlier explored hydrophilic oxidovanadium complexes. Liposomal technology makes oxidovanadium bioavailable, showing enhanced anti-neuroblastoma activity. How is this action against cancer cells achieved? Due to the existence of a highly oxidized environment, vanadium ions are generated in higher cellular concentrations which acts as an effective phosphatase inhibitor leading to fatal cytotoxicity. The delivery of oxidovanadium complexes in vivo for neuroblastoma would likely need more advanced liposomal technology, such as tumour targeting with peptides or antibodies specific for disialoganglioside 2 which is an antigen specific for neuroblastoma cells hence, making it recognisable only by tumour cells. A strategy to prolong the blood circulation time of the liposomes includes the addition of a hydrophilic polymer polyethylene glycol (PEG) moiety onto the surface of the vesicle. Increased liposomal longevity in the bloodstream and reduced immunological responses would reduce drug toxicities. The more targeted and selective delivery of oxidovanadium to tumor tissues would significantly reduce the concerns of off-target toxicities that have beset other oxidovanadium complexes in chronic disease treatment models. The liposomal packaging of oxidovanadium complexes is therefore demonstrated to be a viable approach and should be further tested now in pre-clinical models. Ultimately it would be envisioned for delivery as an intravenous therapeutic, in common with current liposomal formulations. In conclusion, the evidence of good bioactivity of stable, hydrophobic oxidovanadium complexes in neuroblastoma cells is quite strong. Moreover, using liposomal nanotechnology opens up new opportunities for demonstrating the anti-cancer potential of oxidovanadium complexes in pre-clinical cancer models. This would not only be of relevance to neuroblastoma, but also other oxidovanadium-sensitive cancers. If successful pre-clinically, this would strengthen the prospects of generating liposomal formulations for the safe application of oxidovanadium complexes in humans.
Liposomes are small artificial vesicles of spherical shape that can be created from cholesterol and natural non-toxic phospholipids. Due to their size and hydrophobic and hydrophilic character, liposomes are promising systems for drug delivery. These complexes are highly hydrophobic and are able to affect either cytotoxicity or differentiation in several neuroblastoma tumor-derived cell lines. These liposomal particles are more effective than the earlier explored hydrophilic oxidovanadium complexes. Liposomal technology makes oxidovanadium bioavailable, showing enhanced anti-neuroblastoma activity. How is this action against cancer cells achieved? Due to the existence of a highly oxidized environment, vanadium ions are generated in higher cellular concentrations which acts as an effective phosphatase inhibitor leading to fatal cytotoxicity. The delivery of oxidovanadium complexes in vivo for neuroblastoma would likely need more advanced liposomal technology, such as tumour targeting with peptides or antibodies specific for disialoganglioside 2 which is an antigen specific for neuroblastoma cells hence, making it recognisable only by tumour cells. A strategy to prolong the blood circulation time of the liposomes includes the addition of a hydrophilic polymer polyethylene glycol (PEG) moiety onto the surface of the vesicle. Increased liposomal longevity in the bloodstream and reduced immunological responses would reduce drug toxicities. The more targeted and selective delivery of oxidovanadium to tumor tissues would significantly reduce the concerns of off-target toxicities that have beset other oxidovanadium complexes in chronic disease treatment models. The liposomal packaging of oxidovanadium complexes is therefore demonstrated to be a viable approach and should be further tested now in pre-clinical models. Ultimately it would be envisioned for delivery as an intravenous therapeutic, in common with current liposomal formulations. In conclusion, the evidence of good bioactivity of stable, hydrophobic oxidovanadium complexes in neuroblastoma cells is quite strong. Moreover, using liposomal nanotechnology opens up new opportunities for demonstrating the anti-cancer potential of oxidovanadium complexes in pre-clinical cancer models. This would not only be of relevance to neuroblastoma, but also other oxidovanadium-sensitive cancers. If successful pre-clinically, this would strengthen the prospects of generating liposomal formulations for the safe application of oxidovanadium complexes in humans.
Edited by Rachel Cherney
Image Credits:
https://en.wikipedia.org/wiki/Vanadium
https://www.shutterstock.com/video/clip-1016792635-periodic-table-23-vanadium-element-sign-position
Work Discussed:
Irving, E., Tagalakis, A.D., Maeshima, R. et al. The liposomal delivery of hydrophobic oxidovanadium complexes imparts highly effective cytotoxicity and differentiating capacity in neuroblastoma tumour cells. Sci Rep 10, 16660 (2020). https://doi.org/10.1038/s41598-020-73539-6


Leave a comment