Kaye Alcedo
Time is of the essence when it comes to the battle with cancer. Detecting tumors at an early stage increases the chance of patient survival to more than 80%. However, despite today’s advancing technology, about 48% of patients get diagnosed at a very late stage. This is because screening methods of many, if not all, cancers, are still not sensitive enough to detect the tumors. Many patients also do not exhibit symptoms until the cancer has progressed to a more advanced stage (for example – about 60% of liver cancer patients) when the tumor has metastasized to other organs. At this point, the cancer may no longer be amenable to treatment. It is therefore imperative to find new methods of detection.
One such method involves the identification of biomarkers, which are unique molecules that can be found in bodily fluids, such as the blood and urine, and in both normal and diseased tissues. The Foundation for the National Institutes of Health (NIH), recognizing the potential viability of this detection method, partnered up with the Food & Drug Administration and the Pharmaceutical Research and Manufacturers of America to establish the Biomarker Consortium. This partnership’s goal is to discover highly predictive markers that “have an impact on [a] patient’s illness or lifespan”. An ideal biomarker must be detectable when the tumors are present (sensitivity), and must be absent in normal tissue (specificity).
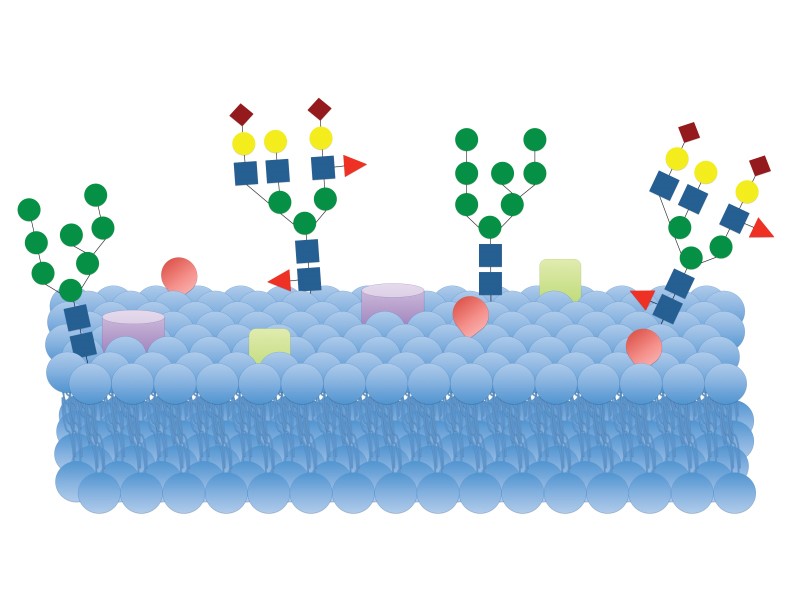
 One example of a biomarker that researchers are currently studying is the glycans connected to proteins. Glycans are sugar molecules that are attached to proteins within the cell. Back in the 1980s, scientists demonstrated that there is a difference between the structures of protein glycans in cancer- compared to normal cells. This led to the discovery that glycans, such as prostate-specific antigen (PSA) in prostate cancer and CD340 in breast cancer, could potentially serve as clinical biomarkers. However, the use of alpha-fetoprotein (AFP) as a biomarker for hepatocellular carcinoma (HCC), which accounts for the majority of liver cancers, is still debated in Europe and the Western Hemisphere. This is because the specificity and sensitivity of AFP vary on the stage of HCC, and the underlying cause of HCC. For example, in patients who end up having HCC, the AFP marker is only present 62.5% of the time (sensitivity), and in patients who do not have HCC, the AFP marker is only absent 53.3% of the time (specificity). These issues are also common in other types of cancers, limiting the efficacy of the current biomarker detection methods. Thus, three decades after the initial discovery of cancer glycans, the NIH allotted specific funding to encourage more research to explain the altered glycans in cancer patients in the hope of discovering more sensitive and more specific biomarkers.
One example of a biomarker that researchers are currently studying is the glycans connected to proteins. Glycans are sugar molecules that are attached to proteins within the cell. Back in the 1980s, scientists demonstrated that there is a difference between the structures of protein glycans in cancer- compared to normal cells. This led to the discovery that glycans, such as prostate-specific antigen (PSA) in prostate cancer and CD340 in breast cancer, could potentially serve as clinical biomarkers. However, the use of alpha-fetoprotein (AFP) as a biomarker for hepatocellular carcinoma (HCC), which accounts for the majority of liver cancers, is still debated in Europe and the Western Hemisphere. This is because the specificity and sensitivity of AFP vary on the stage of HCC, and the underlying cause of HCC. For example, in patients who end up having HCC, the AFP marker is only present 62.5% of the time (sensitivity), and in patients who do not have HCC, the AFP marker is only absent 53.3% of the time (specificity). These issues are also common in other types of cancers, limiting the efficacy of the current biomarker detection methods. Thus, three decades after the initial discovery of cancer glycans, the NIH allotted specific funding to encourage more research to explain the altered glycans in cancer patients in the hope of discovering more sensitive and more specific biomarkers.
Many research groups have started to address this problem. A study of oral cancer showed that there was an overproduction of the protein called DPAGT1 in cancer cells compared to normal, healthy cells. This led to an increase in glycans on a protein that is responsible for maintaining a protective barrier in the oral cavity. Without this barrier, cancerous cells can metastasize and invade other organs, leading to tumor progression. The researchers ultimately concluded that the presence of altered protein glycans in patients with oral cancer was due to another protein that was overactive.
Another group showed that the structural difference in glycans in some cancers is a result of how the sugar molecules are linked to the proteins. Glycans can be attached to the proteins within normal cells via two antennae (imagine the two antennas sticking out of many TVs in the past). In colorectal cancer cells, however, these glycans can be attached via more than two antennae. The connections also contained high amounts of sialic acid, which has been found in higher quantities in metastatic breast cancer. The scientists ultimately found a correlation between the presence of altered glycan structures with a high amount of sialic acid and an overall 41% decrease in 5-year survival rate for patients with colorectal cancer.
In some cases, altered glycans may not be responsible for the progression of cancer cells to highly metastatic and invasive cancer cells. Instead, the normal glycan on a protein may inherently promote cancer development. A study done on hepatocellular carcinoma showed that a specific glycan on the protein CD147 is responsible for transforming normal cells into cancerous cells. Moreover, when the group removed the specific CD147 glycan, the tumors weighed less, appeared smaller, and did not metastasize. They further showed that this removal caused the degradation of the protein. Thus, glycans such as the one on CD147 can not only allow doctors and scientists to identify tumors, but they can also potentially serve as targets for treatment!
Overall, scientists need to further optimize the technology used to detect tumors at early stages of the disease. They have been able to identify differences in protein glycans that are associated with tumors, but the methods they have developed to do so are not yet sensitive or specific enough to be fully effective. Ongoing studies in this particular field are improving our understanding of the mechanisms involved in the alteration of protein glycans. As a result, scientists may find the key to designing more sensitive and specific detection methods for many types of cancer.
Works Discussed
Singal, AG, et al. Detection of Hepatocellular Carcinoma at Advanced Stages Among Patients in the HALT-C Trial: Where Did Surveillance Fail? PMID: 23337478
Strimbu, K. and Tavel, J.A. What are biomarkers? PMID: 20978388
Raedler, A. and Schreiber, S. Analysis of differentiation and transformation of cells by lectins. PMID:3067975
Zhao, J., et al. Protein biomarkers in cancer: Natural glycoprotein microarray approaches. PMID: 19051138
Ahmed Mohammed, HF. And Roberts LR. Should AFP (or any biomarkers) be used for HCC surveillance? PMID: 29085770
Lou, J. et al. Biomarkers for Hepatocellular Carcinoma. PMID: 28469485
Nita-Lazar, M., et al. Overexpression of DPAGT1 Leads to Aberrant N-glycosylation of E-Cadherin and Cellular Discohesion in Oral Cancer. PMID: 19549906
Martin, TA. And Jiang WG. Loss of Tight Junction Barrier Function and its Role in Cancer Metastasis. PMID: 19059202
de Vroome, SW., et al. Serum N-glycosme Alteration in Colorectal Cancer Associate with Survival. PMID: 30093973
Bhide, GP. And Colley, KJ. Sialylation of N-glycans: mechanism, cellular compartmentalization and function. PMID: 27975143
Teoh, ST., et al. Sialic Acid Metabolism: A Key Player in Breast Cancer Metastasis Revealed by Metabolomics. PMID: 29892572
Li, JH. et al. N-linked glycosylation at Asn152 on CD147 affects protein folding and stability: promoting tumour metastasis in hepatocellular carcinoma. PMID: 27869218
Image Credits
Recreated by the author based on the original figure in Glycosylation Changes in Cancer