Reading time: 5 minutes
Colette Bilynsky
The American Cancer Society has found colorectal cancer to be the 3rd highest cause of cancer deaths and 3rd highest estimated new cases in both men and women in 2022 [1]. The risk of colorectal cancer is significantly higher in people with inflammatory bowel disease (IBD): ulcerative colitis (UC), and Crohn’s disease. Furthermore, patients with a history of these types of gastrointestinal issues tend to have a worse prognosis than those without [2]. These statistics demonstrate a correlation between these types of inflammatory intestinal conditions and colorectal cancer. This begs the question: how do inflammatory bowel diseases cause cancer?
The answer is a bit uncertain, however, data suggest that inflammation is the main culprit. Ulcerative colitis is a type of IBD that inflames the digestive tract, causing painful sores on the innermost lining of the rectum and colon. Crohn’s disease, meanwhile causes inflammation in any part of the digestive tract from a person’s mouth to anus, which affects the entire bowel wall thickness, as opposed to just the innermost layer [3]. A meta-analysis (or a study that systematically combines the results of multiple independent studies) found that individuals with UC were more than 2 times more likely to develop CRC [4], and a Scandinavian study found that individuals with Crohn’s disease had a similar increased risk of cancer [5]. When researchers looked to patients with IBD who eventually developed a type of CRC, called Colitis-Associated Colorectal Cancer (CAC), they found that the level of inflammation in these patients was a risk factor for CAC development [2].
Patients with IBD have a level of constant inflammation that causes their digestive symptoms [6]. But what does that actually mean? You might think of inflammation as the swelling that occurs around an infected wound, but within your body inflammation is a cascade of events that help your cells fight infections. Typically, this includes your immune cells going to the infected area to help fight the infection, helped by the release of cytokines (a type of proteins that cells release to tell other cells or themselves what to do) and increased blood flow to that area (which is what causes the swelling and heat associated with inflammation) [7]. However, in the case of IBD, the inflammation is chronic (long lasting) and not just occurring in response to an infection or foreign stimulus, which can have negative effects on the body [2,6].
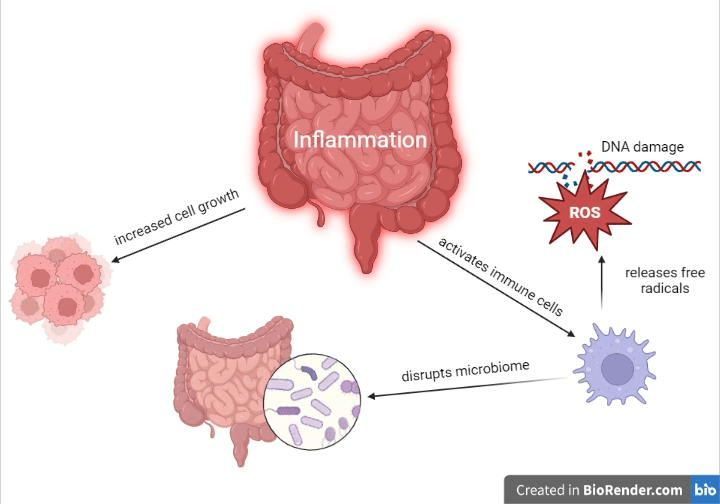
One of the major negative effects of inflammation is genetic mutations, which can cause cancer. During inflammation, activated cells release molecules called free radicals that damage DNA. When fighting bacteria, this is a good thing, as DNA damage can kill the invading bacteria [7]. However, constant free radical release in the colon, as is the case in IBD, can cause genetic changes to the patient’s epithelial cells lining the colon [2]. The inflammation promotes increased cell growth, which means that these mutated cells are then encouraged to replicate more rapidly. Lastly, the inflammation affects the gut’s microbiome [2]; your digestive system has a complex microbiome made up of different bacterial types that help digest your food and keep you healthy. In individuals without IBD, pro- and anti-inflammatory signals are kept in check so that your immune cells can tolerate the bacteria of the microbiome[2]. As explained above, inflammation primes your immune system to kill bacteria and other foreign intruders, and in the case of patients with IBD, the balance in your digestive system leans towards more inflammatory, attacking the microbiome [2]. When this microbiome is disturbed, the epithelial layer can be disrupted and harmful reactive metabolites (molecules created as the result of the body digesting or otherwise breaking down some kind of substance) can be released. All of these factors working together create a favorable environment for cancer to develop [2].
Understanding this link between inflammation and colorectal cancer can help researchers and clinicians identify therapies that IBD patients can take to prevent cancer. 5-ASA, sometimes called mesalazine, is a nonsteroidal anti-inflammatory drug (NSAID) like aspirin that is sometimes taken by patients during UC flare ups and is believed to reduce tumor development [2]. Besides its anti-inflammatory effects, it can help reduce the amount of DNA mutations and activates PPAR-y. PPAR-y is a transcription factor that causes the expression of a family of hormone receptors that regulate cell growth and death within the colon, suggesting a mechanism for its tumor-reducing property [8]. However, there have been some conflicting results when 5-ASA’s ability to prevent CRC was tested, with some studies finding that it does decrease the risk of developing CAC and some saying that there was no effect [9]. Another potential therapeutic would be anti-TNF therapies. TNF-α is a type of cytokine that cause pro-inflammatory activation, and has been shown to be involved in CAC development specifically [2]. Anti-TNF therapies belong to the biologic class of drugs, and are usually monoclonal antibody treatments. The antibodies your body naturally makes target substances foreign to your body, like bacteria and viruses. For monoclonal antibody treatments, researchers design the antibody to target a specific protein, in this case, either TNF-α or its receptor TNFR, this prevents them from binding and activating the associated pro-inflammatory pathway. Anti-TNF therapies are already sometimes prescribed for the management of UC and Crohn’s disease, and have been shown in animal models to prevent CAC [10]. However, further clinical trials in humans are needed to confirm this anti-cancer effect in patients [2,9]. While researchers work to find the best ways of preventing CRC, it is vitally important that at-risk individuals continue to be regularly screened so that any tumors can be found early and treated.
Edited by Maha Said
Works Cited:
[1] “Cancer Facts & Figures 2022| American Cancer Society.” https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2022.html (accessed Feb. 26, 2023).
[2] M. C. Fantini and I. Guadagni, “From inflammation to colitis-associated colorectal cancer in inflammatory bowel disease: Pathogenesis and impact of current therapies,” Digestive and Liver Disease, vol. 53, no. 5, pp. 558–565, May 2021, doi: 10.1016/j.dld.2021.01.012.
[3] “What Is Crohn’s Disease?,” Crohn’s & Colitis Foundation. https://www.crohnscolitisfoundation.org/what-is-crohns-disease (accessed Feb. 26, 2023).
[4] T. Jess, C. Rungoe, and L. Peyrin–Biroulet, “Risk of Colorectal Cancer in Patients With Ulcerative Colitis: A Meta-analysis of Population-Based Cohort Studies,” Clinical Gastroenterology and Hepatology, vol. 10, no. 6, pp. 639–645, Jun. 2012, doi: 10.1016/j.cgh.2012.01.010.
[5] O. Olén et al., “Colorectal cancer in Crohn’s disease: a Scandinavian population-based cohort study,” The Lancet Gastroenterology & Hepatology, vol. 5, no. 5, pp. 475–484, May 2020, doi: 10.1016/S2468-1253(20)30005-4.
[6] D. C. Baumgart and S. R. Carding, “Inflammatory bowel disease: cause and immunobiology,” The Lancet, vol. 369, no. 9573, pp. 1627–1640, May 2007, doi: 10.1016/S0140-6736(07)60750-8.
[7] K. Murphy and C. Weaver, Janeway’s Immunobiology, 9th ed. Garland Science, 2017.
[8] C. Rousseaux et al., “Intestinal antiinflammatory effect of 5-aminosalicylic acid is dependent on peroxisome proliferator–activated receptor-γ,” Journal of Experimental Medicine, vol. 201, no. 8, pp. 1205–1215, Apr. 2005, doi: 10.1084/jem.20041948.
[9] A. Lopez, L. Pouillon, L. Beaugerie, S. Danese, and L. Peyrin-Biroulet, “Colorectal cancer prevention in patients with ulcerative colitis,” Best Practice & Research Clinical Gastroenterology, vol. 32–33, pp. 103–109, Feb. 2018, doi: 10.1016/j.bpg.2018.05.010.
[10] B. K. Popivanova et al., “Blocking TNF-α in mice reduces colorectal carcinogenesis associated with chronic colitis,” J Clin Invest, vol. 118, no. 2, pp. 560–570, Feb. 2008, doi: 10.1172/JCI32453.


Loved reading this post.
Very informative article that sheds light on the link between inflammatory bowel diseases and colorectal cancer. The potential therapeutic options mentioned for preventing cancer in at-risk individuals are also intriguing.
All the best
Frank
meweightloss.com
LikeLike