Reading time: 3 minutes
Hannah Young
Somatic mutations are changes in DNA that occur as cells in our body divide. They are distinct from germline mutations, which occur in germ cells (e.g. eggs and sperm) and are passed down from parent to child. Both somatic and germline mutations contribute to cancer and aging. While we’ve learned a lot about the types and frequency of mutations that occur in human DNA, we know much less about mutations in non-human animals, largely due to a lack of available samples to study.
However, more recent efforts focused on studying the prevalence of cancer and the cancer mortality risk across non-human animals have given us new insight. It has long been hypothesized that larger and longer-lived animals are at greater risk for cancer because they have a greater number of cells that undergo more rounds of cell division, ultimately creating more opportunities for mutations to occur (and potentially cause cancer) throughout their lifespan. However, in reality, we’ve observed that larger, longer-lived organisms, such as elephants are overall less likely to get cancer than smaller, shorter-lived animals. This mismatch between probability and outcome is known as Peto’s Paradox, and several studies have revealed the biological culprits behind it. Elephants, for example, have been found to have 20 copies of the tumor suppressor gene TP53, while humans only have one.
More recently, a study focusing on measuring mutation rates across mammals added another piece to Peto’s paradox puzzle. Mutation rate, or the frequency at which mutations occur in different cells, tissues, etc., is affected by both cell-intrinsic factors, such as flaws in the machinery that corrects errors in DNA replication during cell division, and extrinsic factors, such as environmental carcinogens like UV and cigarettes. Cagan et al. studied 16 mammalian species to capture differences in mutation rates in mammals and determine what characteristics, including what types of mutations, lifespan, and body size, might contribute to these differences.
To focus on intrinsic factors affecting mutation, the authors studied the mammals’ intestinal crypts or cells that line the small intestine and colon, which are thought to originate from a single stem cell and are resistant to environmental carcinogens. Through sequencing DNA from crypt cells across individuals of different ages from different species, the authors caught a clear correlation between the total number of mutations and the individual’s age, suggesting that somatic mutations accumulate with age.
Digging deeper into their sequencing, the authors also wanted to know if the same DNA damage repair mechanisms causing mutations were the same across mammals. Identifying signatures of different types of mutations could help identify which DNA damage repair pathways are responsible for the mutations observed across species. While three mutation-causing processes were dominant across all species, their contribution to each individual species was varied.
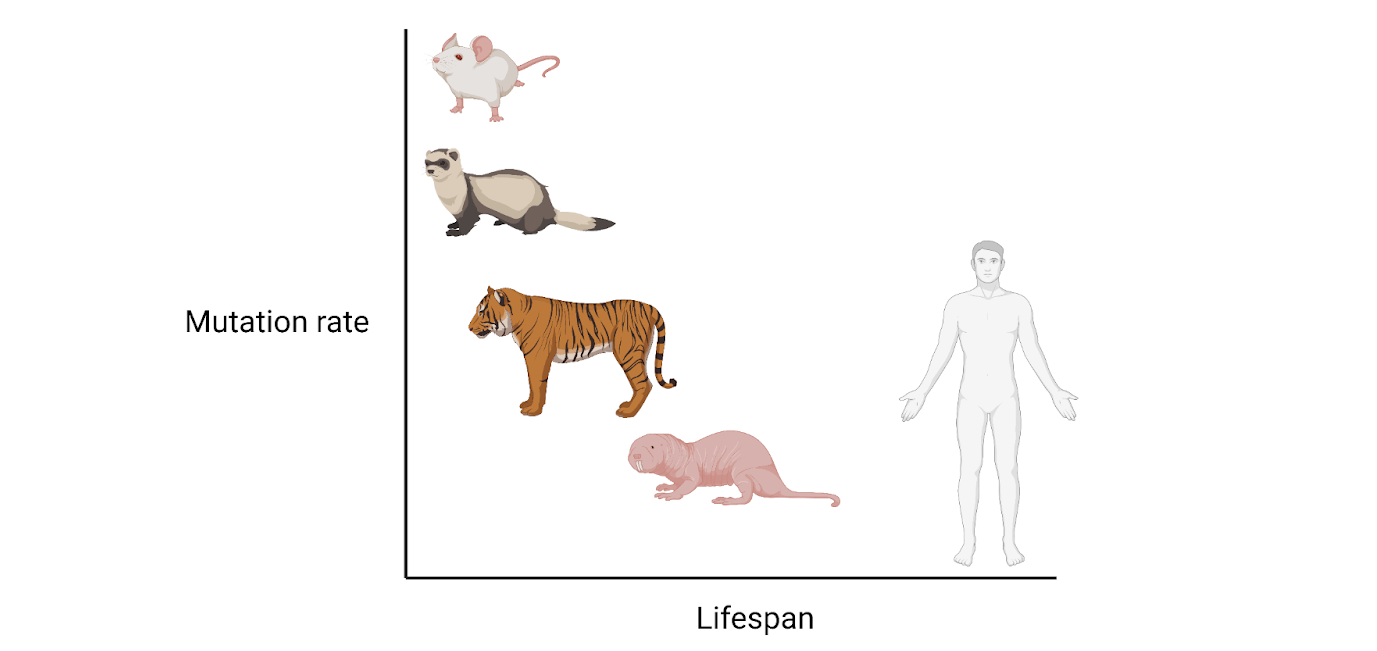
Lastly, the authors wanted to know if the somatic mutation rate was associated with any life-history traits, such as body size or lifespan. Using lifespan data from zoos to minimize extrinsic effects on lifespan (such as predation, food availability, etc), the authors found a remarkably strong correlation suggesting that mammals with longer lifespans have lower mutation rates. Larger body mass also correlated with fewer somatic mutations, though more weakly and with less statistical confidence. The importance of the connection between lifespan and mutation rate was highlighted by two species, giraffe and naked mole-rat, which have similar lifespans and mutation rates, but vastly different body sizes.
Somatic mutations drive cancer and other diseases associated with aging. However, because mutations and the biological processes they affect have been largely studied in humans, we’ve known little about their occurrence in non-human animals. This study highlights the importance of comparative analyses across animals in understanding health and disease and addresses the long-standing questions in the field by investigating the basic principles of how and how often mutations occur.
Edited by Emily Costa
Work discussed
Cagan, A., Baez-Ortega, A., Brzozowska, N. et al. Somatic mutation rates scale with lifespan across mammals. Nature 604, 517–524 (2022). https://doi.org/10.1038/s41586-022-04618-z

Leave a comment