Reading time: 4 minutes
Anthony (Tony) Tao
Controlling the genes encoded by our DNA has always been a dream in oncology. Just as our healthy cells rely on genes to properly function, cancer cells depend on our genes to thrive and parasitize our bodies. In the late 90s, a powerful technology was developed called RNA interference (RNAi), which has the power to precisely control DNA function. This technique relies on RNA, a multifaceted molecule similar to DNA, and allows scientists to abolish expression of specific genes encoded in DNA.
In the 2000s following its advent, RNAi was the therapeutic vogue. A spate of pharmaceutical companies began investing time and money into developing RNAi-based therapies, especially for cancer, which are often vulnerable to genetic disruptions. However, as the first decade of the 21st century neared its end, so did excitement in RNAi. The flurry of RNAi-focused clinical trials that had been initiated were met with disappointment. The prevailing issue was the inefficient delivery mechanisms for bringing RNA into target cells. This was especially true for cancers that reside in the brain, which is guarded by the blood-brain barrier (BBB), a structure infamously discriminatory about what it allows into the brain. Recently, advances in RNA packaging and delivery have led to resurrected interest in RNAi as a therapeutic tool. Notably, a research group at Northwestern University presented convincing evidence that RNAi can be a tool in treating glioblastoma multiforme in human patients.
Arguably one of the most lethal human cancers, glioblastoma multiforme (GBM) is both terrifyingly aggressive and difficult to treat. It is a fast-growing tumor that arises from helper cells in the brain called astrocytes. Patients with GBM can experience a wide array of neurological symptoms such as vision impairments, headaches, personality changes, and seizures. Moreover, because of its residence in the brain, GBM is uniquely shielded by the BBB from many types of drugs designed to kill tumors.
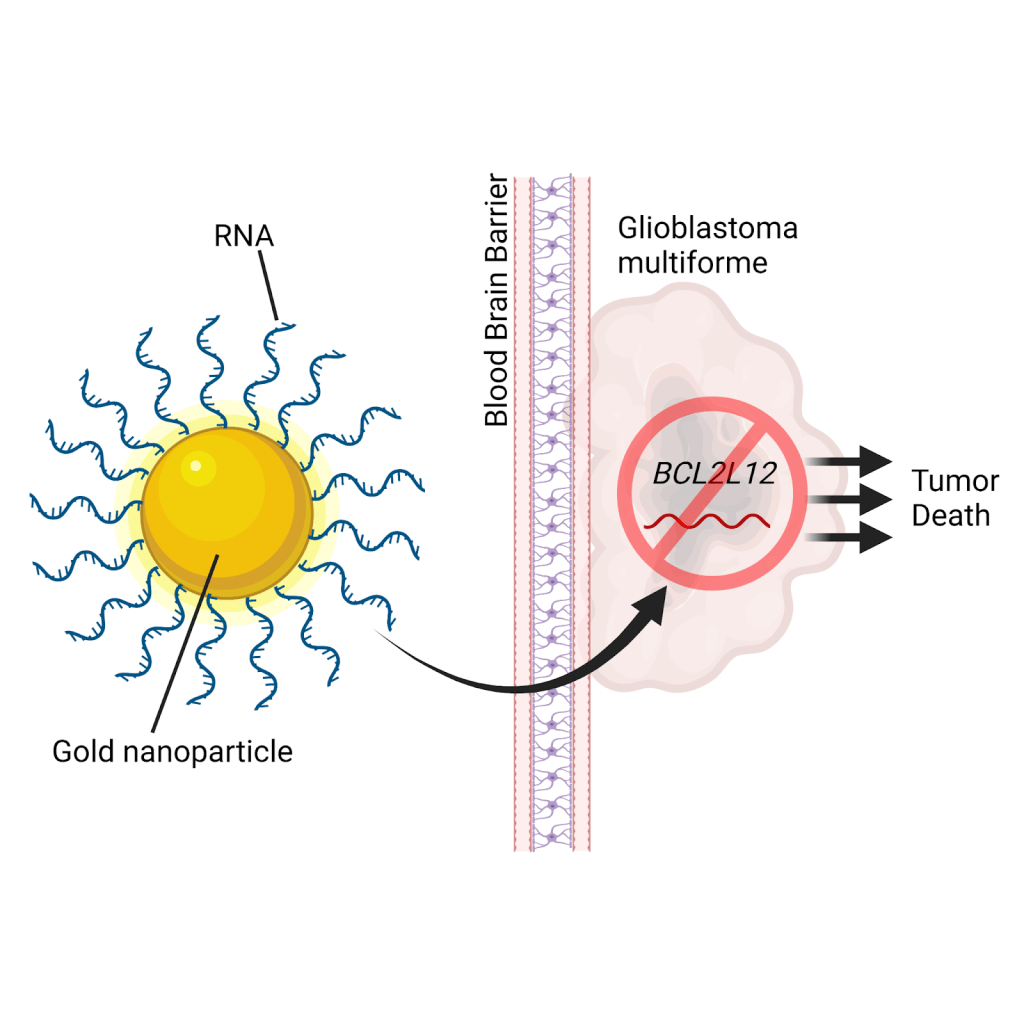
In 2013, researchers at Northwestern developed a novel delivery method with which RNAi can be targeted to brain tumors and ultimately modulate their genes. Specifically, they coated gold nanoparticles with RNA molecules designed to specifically silence the gene BCL2L12, on which GBM depends to sustain growth. When the researchers injected these RNA-studded nanoparticles into mice bearing brain tumors, they saw that these RNA particles crossed the BBB and accumulated in the tumor cells. They also found that treatment diminished the levels of BCL2L12 as intended and ultimately delayed cancer progression in brain-tumor-bearing mice. At this point, however, it remained unknown whether such a treatment could be effective in human GBM.
More recently in 2021, these same scientists presented powerful evidence that their RNA nanoparticles could have real clinical benefits in humans. First, they injected the RNA particles into monkeys and confirmed that the treatment is safe. Next, they enrolled 8 human patients with GBM for a Phase 0 clinical study. The main goal was to determine if the RNA nanoparticles, upon injection, can overcome the brain’s natural barricade and reach the tumor. Shortly after injection, the patients underwent necessary surgery to remove the GBM tumor. Excitingly, by analyzing these resected tumor samples, the researchers found that the nanoparticles successfully entered the brain and accumulated in the tumor. Remarkably, in 2 of the patients who needed a second surgery months later, the RNA particles continued to persist in the tumor, suggesting this treatment may have long-lasting effects. Since it was a Phase 0 trial, the researchers were only permitted to treat these patients with very low doses of the particles, lower than the dose required for effective clinical treatment. Despite this, the reduced quantity of RNA nanoparticles found in the tumor were still capable of reducing the intended gene target: tumor-promoting BCL2L12.
Never before has the prospect of leveraging RNAi to attack human brain tumors been so closely realized. The power of the RNA nanoparticle technology lies not only in its capacity to reach the brain, but also in its potential versatility. In the Phase 0 study, the scientists focused only on one genetic target ‒ BCL2L12. But, by simply modifying the RNA code on these nanoparticles, other genes could also be potentially targeted, perhaps even multiple genes simultaneously. These approaches can thus be uniquely tailored to swiftly kill all types of tumors ‒ whether they are tumors of the brain or beyond.
Edited by Gabrielle Dardis


Leave a comment