Keighley Reisenauer
Reading time: 3 minutes
Think back to 2003, when Jamie Lee Curtis and Lindsay Lohan swapped bodies as mother and daughter in the flick Freaky Friday. While their bodies looked the same, their behaviors changed as each brought their own personality to the other’s lives. Movies love this idea of swapping bodies, brains, or souls. It’s the stuff of fantasy and science fiction. However, it isn’t truly as far off as the entertainment industry would lead you to believe.
Now, think back to high school biology where you learned about the different parts within a cell: the nucleus, the ribosomes, the Golgi, and the mitochondria (here is where you all internally yell “the powerhouse of the cell!”). You may recall that the nucleus is the “brain” of the cell– it carries the DNA responsible for coding proteins that tell each cell what it is and what it should do. It directs internal traffic and responds to environmental signals. And, in 1994, scientists successfully completed the first cellular “brain transfer” [1]. Briggs and King began by enucleating cells by removing their nucleus, leaving an empty shell awaiting instructions. Then, they transplanted the nucleus of a blastula cell into the enucleated eggs of frogs, which caused tadpoles to hatch normally. By taking the “brain” of a cell that is undergoing development and putting it into a dormant egg cell, it was sufficient to initiate and complete the developmental process. Since then, scientists have explored the bounds of this cellular “brain transplant” from the 1996 Dolly the Sheep to stemness as regenerative medicine to re-wiring cancer cells. In the case of stem cell research, enucleation is helpful when donor stem cells can be created to more accurately match a patient, especially in the cases of creating tissues or organs.
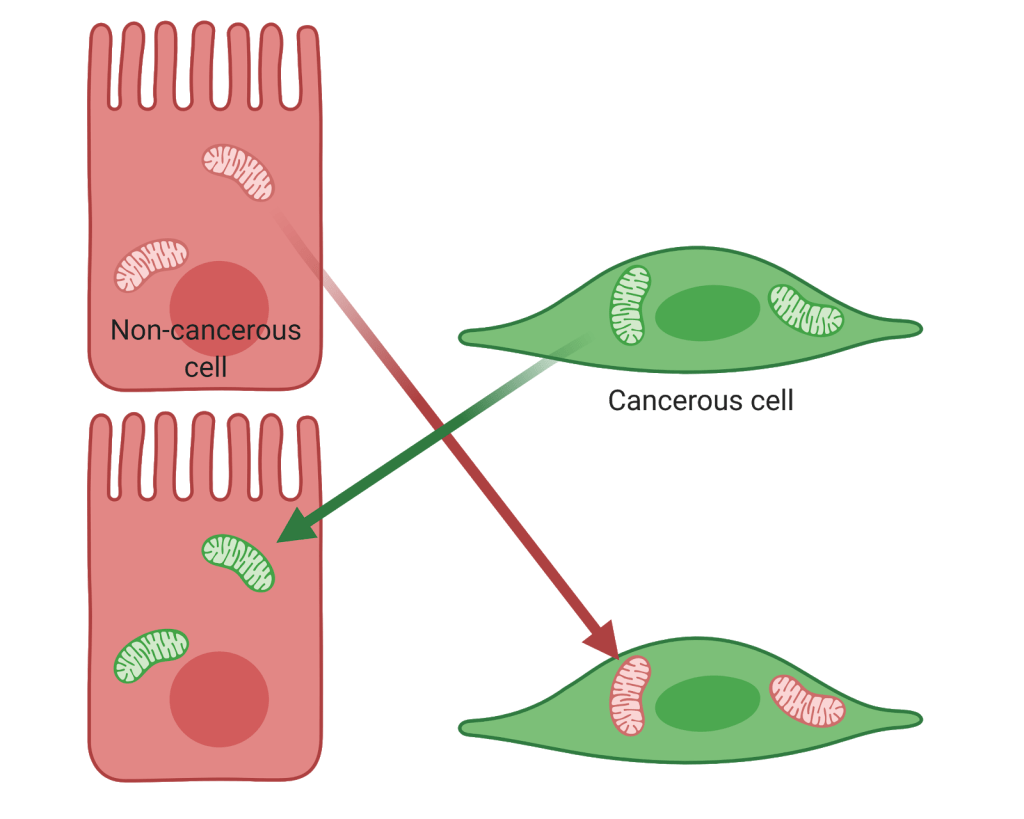
With the advancement of science inherently comes the deepening of questions and exploration. It is known that the nucleus is not the only organelle to contain DNA; in fact, mitochondria also carry 37 genes of their own [2]. Scientists began to wonder if changes in mitochondria could impact a cell. A 1998 study demonstrated that, in colorectal cancer, mutations linked to the cancer were found in the mitochondrial DNA [3]. This suggested that mitochondrial changes could play a significant role in impacting the behavior of the cell. Then, in 2013, a group of scientists from Baylor College of Medicine definitively linked the impact of the mitochondria to cancer cell behavior [4]. Mitochondria from aggressive, metastatic osteosarcoma cells were swapped with mitochondria from benign breast cancer cells in a process termed “transmitochondrial cybridization.” These mitochondria-swapped cells, termed cybrids, contained a native nuclear background and the only change was in the mitochondria. The presence of the benign mitochondria reversed cancerous characteristics including cell proliferation, viability under hypoxia, resistance to apoptosis (cell death), resistance to anti-cancer drugs, and invasion. Further, upon investigation of the genetics, many oncogenic pathways were inhibited with the presence of non-cancerous mitochondria. Right now, this process is conducted using cell lines in laboratory research, but the hope is that it can be further studied to be used clinically. Drawing on our understanding from enucleation experiments for patients, the relevance of the mitochondria as it influences the nucleus may need to be considered when preparing tissues or organs from donors.
The implication of these findings and others like it [5] solidify not only the importance of the role of the mitochondria, but also the critical influence of the mitochondria on the nucleus. This opens the door to regulating the mitochondrial-nuclear cross talk as a promising target in cancer therapy.
Edited by Rachel Cherney
Primary Article:
Kaipparettu, B. A. et al. Crosstalk from non-cancerous mitochondria can inhibit tumor properties of metastatic cells by suppressing oncogenic pathways. PLoS ONE 8, e61747 (2013).
Images:
Waters, M. (2003). Freaky Friday. Buena Vista Pictures.
(BioRender, created by K. Reisenauer)
References
- Collas P, Barnes FL. Nuclear transplantation by microinjection of inner cell mass and granulosa cell nuclei. Molecular reproduction and development. 1994 Jul;38(3):264-7.
- Chial H, Craig J. mtDNA and mitochondrial diseases. Nature Education. 2008;1(1):217.
- Polyak K, Li Y, Zhu H, Lengauer C, Willson JK, Markowitz SD, Trush MA, Kinzler KW, Vogelstein B. Somatic mutations of the mitochondrial genome in human colorectal tumours. Nature genetics. 1998 Nov;20(3):291-3.
- Kaipparettu, B. A. et al. Crosstalk from non-cancerous mitochondria can inhibit tumor properties of metastatic cells by suppressing oncogenic pathways. PLoS ONE 8, e61747 (2013).
- Imanishi H, Hattori K, Wada R, Ishikawa K, Fukuda S, Takenaga K, Nakada K, Hayashi JI. Mitochondrial DNA mutations regulate metastasis of human breast cancer cells. PloS one. 2011 Aug 11;6(8):e23401.
Other Resources/Further Reading:
- Chen, H. & Chan, D. C. Mitochondrial dynamics in regulating the unique phenotypes of cancer and stem cells. Cell Metab 26, 39–48 (2017).
- Márquez-Jurado, S. et al. Mitochondrial levels determine variability in cell death by modulating apoptotic gene expression. Nature Communications 9, 389 (2018).
- Ubah, O. & Wallace, H. Cancer Therapy: Targeting Mitochondria and other Sub-cellular Organelles. Current Pharmaceutical Design 20, 201–222 (2014).
- Zhao, J. et al. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene 32, 4814–4824 (2013).


Leave a comment