Reading time: 4 minutes
Suchitra Mitra
Majority of cancer associated deaths occur from metastasis, when cells from the primary tumor migrate to a distant site within the body. The migration of cancer cells usually occurs via three routes — direct invasion of surrounding tissues, the circulatory system (ie, blood), and the lymphatic system. Recent developments have revealed a fourth route of migration, via the nervous system.
Neurogenesis refers to the formation of new functional neurons from neuronal precursors. Neurogenesis has been reported to be associated with various solid tumors — almost all pancreatic cancers, 80% of head and neck cancers, 75 % of prostate cancer, and 33% of colorectal cancer, and these are also associated with increased cancer proliferation and metastasis. Tumor associated nerve fibers invade the central nervous system and cause neuropathic pain and side effects. In addition to neurogenesis, tumors drive axono-genesis (enlargement of nerves and increase in nerve density) and neural reprogramming (differentiation of nerves to different varieties). These processes promote tumor survival and invasion in animal models of cancer. Hence, the development of cancer and its subsequent metastasis is supported by the development of a nervous system, in addition to an immune and vascular network around the tumor.
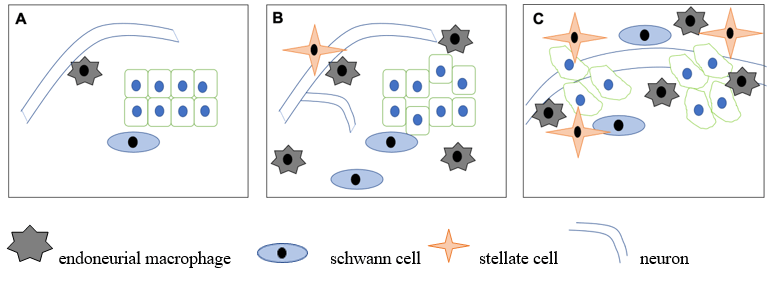
The process by which cancer cells recruit nerve fibers, to facilitate cell-cell communication that serve as tracking guides for migration, is called perineural invasion (PNI). PNI in cancer is a multifaceted process with several contributing factors. Perineural niche — a microenvironment composed of several components including neurons, supporting cells, inflammatory cells, blood vessels, and signaling molecules leads to nerve cell-cancer cell cross talk and contributes to the PNI invasion pathway. The nerve growth patterns towards the tumors are diverse and PNI can exist as an independent mode of invasion even if there is no lymph or blood invasion. Similar to the micro environment around a cancer cell, lower oxygen levels, high glucose levels, inflammation, and sympathetic system activation form the microenvironment around PNI and also acts as a driving force for PNI.
Cancer cells invade nerve cells by inducing a cascade of inflammation, wound healing, and anti-apoptosis processes. The seven hallmarks of neural tracking in cancer cells are — inflammatory response, cancer cell metastasis towards nerves, neurogenesis, cancer cell adhesion to nerve sheaths, cancer cell survival, nerve homeostasis, and nerve invasion. Cancer cells recruit endoneurial macrophages, immune cells responsible for cellular defense and regeneration of peripheral nerves, to produce growth factors for invasion in prostate cancer and in the highly malignant pancreatic ductal adenocarcinoma (PDAC)—an example of how inflammatory response in PNI plays a role in metastasis.
Cancer cells also activate stellate cells, which produce the protein galectin-1 and activate signaling molecules that induce metastasis. Additionally, cancer also activates Schwann cells, the most prevalent cell type in peripheral nerves, towards PNI initiation with the help of brain derived nerve growth factors and tropomyosin receptor kinase B protein. Cancer cells also hijack neurotransmitters, chemokines, and other neurotrophic growth factors, and chemotactic molecules to drive metastasis. Nerve injury within the perineural niche drives neural homeostasis and regeneration and itself acts as a portal of invasion. DNA damage in neurons leads to more aggressive cell growth and suppresses apoptosis.
Initially, PNI was believed to be an analogous pathway of lymphatic invasion. Also, PNI was considered a low resistance pathway for tumor spread. However, recent discoveries establish PNI as a highly directed process with reciprocal interactions between cancer and the nervous system of the host. The path of migration around nerves is also established as high resistance because of the presence of multiple layers of collagen in the neuron sheath. The limitations in understanding PNI mechanisms lie in the lack of established experimental methods to study the complex interactions of the nerve and cancer cell. PNIs are also underreported in different cancers. Currently the definition of PNI involves the presence of cancer cells inside nerves or outside nerves within 33% of nerves periphery and scientists have just begun to scratch the surface of PNI’s evolving pathogenesis.
The molecular mechanisms associated with PNI and nerve cell-cancer cell crosstalk is still not well understood, and it embodies the potential for novel therapeutic avenues. With deeper understanding of the nerve-cancer cross talk, novel anti-neurogenic drugs can be developed that can be used together with conventional chemotherapies and immunotherapies.
Edited by Manisit Das
Works Discussed
1. Amit, M., Na’ara, S. & Gil, Z. Mechanisms of cancer dissemination along nerves. Nat Rev Cancer 16, 399–408 (2016). https://doi.org/10.1038/nrc.2016.38
2. Cervantes-Villagrana, R.D., Albores-García, D., Cervantes-Villagrana, A.R. et al. Tumor-induced neurogenesis and immune evasion as targets of innovative anti-cancer therapies. Sig Transduct Target Ther 5, 99 (2020). https://doi.org/10.1038/s41392-020-0205-z
3. Bapat AA, Hostetter G, Von Hoff DD, Han H. Perineural invasion and associated pain in pancreatic cancer. Nat Rev Cancer.11, 695 (2011). doi: 10.1038/nrc3131. PMID: 21941281.
4. Kuol, N., Stojanovska, L., Apostolopoulos, V. et al. Role of the nervous system in cancer metastasis. J Exp Clin Cancer Res 37, 5 (2018). https://doi.org/10.1186/s13046-018-0674-x

Leave a comment