When patients volunteer to participate in a clinical trial, their voices should be heard.
Reading time: 7 minutes
Patty Spears
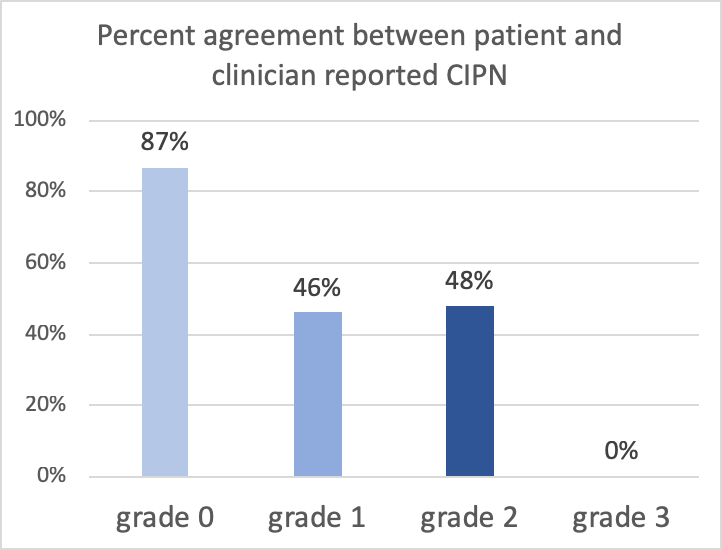
A recent study (Nyrop et. al. 2019) looked at how clinicians and patients reported numbness and tingling in the patient’s hands and feet (peripheral neuropathy) after getting chemotherapy for breast cancer. They call this chemotherapy induced peripheral neuropathy (CIPN). They found differences in reporting the severity of CIPN by patients compared to clinicians. They found the largest differences in reporting when symptoms of CIPN were most severe. This suggests that clinicians may be under-reporting the severity of CIPN when compared to patients reporting their own symptoms when CIPN is most severe. CIPN is a common and potentially debilitating, long lasting side effect of some chemotherapy treatments that can negatively affect a patient’s quality of life. This example supports the need for both patient and clinician reporting of side effects, especially in the case of CIPN.
Hearing the Patient Voice through Patient Reported Outcome Measurements (PROMs)
In developing appropriate PROMs for a specific patient population, consideration for what matters to the patient should always be placed at the top of the list.
Patient reported outcome measurements (PROMs) are described as any report of the status of a patient’s health condition that comes directly from the patient, without interpretation by a clinician or anyone else. One way patients can report how they are feeling (their health status) is by filling out a questionnaire. Over many years, there have been different types of questionnaires developed and tested for research. The choices are vast, but sometimes more is not necessarily better. For example, a questionnaire may be given to a patient that might not ask appropriate questions about what the patient is experiencing at that time. However, there is general agreement to use patient questionnaires to measure the patient experience in clinical trials. A fellow patient advocate once stated, “PROMs should measure not only what is the matter with the patient but what matters to the patient”. In developing appropriate PROMs for a specific patient population, consideration for what matters to the patient should always be placed at the top of the list.
Patient reported outcome measurements (PROMs) are not new; they have been around since the 70’s with the first introduction of quality of life (QOL) questionnaires (Patterson, 1975; van Dam, 1981). Due to the broadness of the QOL measures, they have been refined over time with the introduction of health-related quality of life (HR-QOL) questionnaires in the 90s (Wilson IB, JAMA 1995, 273). Although these are more focused on the patient experience, the current HR-QOL measurements are still broad and ask many questions that may not be meaningful to patients.
There has been a recent effort to further refine PRO questions (Kluetz, 2016) to measure what is most relevant and meaningful to patients. Kluetz et. al. proposed a new model to ask thoughtful questions that directly measure what patients are experiencing as a result of their treatment. These core questions include the effect of the therapy on a patient’s symptoms (disease symptoms), side effects from the therapy (symptomatic adverse events) and the effect of the therapy on the normal activity of the patient (physical function). By concentrating questions on areas more likely to be related to the treatment and more likely to be important to patients, there are new opportunities to refine PRO measurements. For example, the development of patient reported symptom monitoring (PRO-CTCAE®; Basch 2014), is a step in the right direction. PRO-CTCAE® has individual symptom questions used by themselves or in any combination meaningful to a specific patient population. This is very different from using a questionnaire with many questions that must be used in its entirety. Unfortunately, not all of the questions are always relevant and meaningful to a specific patient population.
Making the Case for using PROMs in Clinical Trails
Clinical trials are research studies involving human volunteers. Clinical trials test new treatments in an orderly way to ensure safety while determining whether a treatment is effective. Although most of the focus is on testing the treatment for how well it works (efficacy) and harms it may cause (safety), it is just as important to measure how the patient feels and functions while receiving the treatment. PROMs will describe how patients are feeling and functioning when comparing the new treatment to usual care.
A recent article by Zettler et al (2019) highlights the need for PROMs in clinical trials. They looked at the use of PRO questionnaires in studies submitted for FDA drug approval between 2011 and 2017. They found that only 70% (50 of 71) collected PROMs in pivotal trials supporting initial approval. The importance of PROMs has increased because of these 71 trials, only 32% (23 of 71) were approved based on increasing how long the patient lived (overall survival). This means most approved drugs did not show a survival advantage, but used other endpoints for approval. These endpoints are used as an indicator or sign in place of survival to determine if a treatment works. The problem is that without an indication that the patient lives longer (survival) or better (PROMs), it is unclear if any of these endpoints are a true indication of how well the treatment works and if it is meaningful to patients. For example, looking at whether a tumor gets smaller does not tell you if the patient feels better or lives longer. If patients do not live longer, it is important to know whether patients benefit by living better. For this reason, it is critical to include PRO questionnaires in all clinical trials to determine the true meaningfulness and value to patients.
Patients want to know how other patients tolerated a new treatment compared to the usual treatment. With new treatment options, patients are challenged to weigh the potential benefits against the potential harms. Many times patients are given a choice, and decisions need to be made with accurate and meaningful information that matters to patients. Unfortunately, the information from other patients about how they felt and functioned on a new treatment is not always available. As discussed earlier (Nyrop 2019), relying only on clinicians reporting of how patients feel and function may not be accurate due to under-reporting. Additionally, many trials do not show that patients live longer, so there is a greater need to know if patients live better (Zettler, 2019).
Challenges and Recommendations
PRO questionnaire use in clinical trials has been ongoing for many years, but there have been challenges. Even though patients have been advocating for the use of PRO questionnaires in clinical trials there is still reluctance to include PROMs. Unfortunately, when included there is minimal planning to make sure the questions are appropriate and the research question is important to patients. Although there are some real barriers to including PROMs in every clinical trial, it is important to change the image of PROMs and treat them as a valuable and necessary measurement.
In the end, it is vital to include patients in discussions about including the right PRO questionnaires in clinical trials. Using patient informed PRO questionnaires means asking the right question, at the right time, in the right way. This will lead to the most meaningful information for patients and will create an environment for open communication between clinicians and their patients. PROMs can inform future patients about current patient experiences so they know the true overall benefits and harms of a new treatment.
| Challenges | Recommendations |
| One size fits all approach | Develop targeted (precision) PROMs asking questions that matter to patients. |
| Broad questionnaires asking questions not proximal to the patient experience | Ask what matters to patients based on core elements: disease symptoms, treatment side effects, physical function. |
| PROMs not usually a primary or secondary objective in clinical trials | Develop PROMs with rigor and treat them as an important part of the trial answering specific questions as a primary or secondary objective. |
| Analysis and reporting of PROMs done later and published in different journals, usually behind paywalls. | Analyze and report PROMs as part of the primary clinical trial publication, and make them accessible. |
| Patients are not given feedback regarding the PROMs collected in clinical trials. | Include feedback to the patient as part of the clinical trial. |
| Too much missing data | Let patients know why the questions are being asked and their importance. Make questions relevant and use a method and timing that are acceptable to patients. |
Patty Spears is a Patient Research Advocate and Scientific Research Manager at the Office of Clinical and Translational Research, UNC Lineberger Comprehensive Cancer Center.
References:
Nyrop KA, Deal AM, Reeder-Hayes KE, Shachar SS, Reeve BB, Basch E, Choi SK, Lee JT, Wood WA, Anders CK, Carey LA, Dees EC, Jolly TA, Kimmick GG, Karuturi MS, Reinbolt RE, Speca JC, Muss HB1. Patient-reported and clinician-reported chemotherapy-induced peripheral neuropathy in patients with early breast cancer: Current clinical practice. Cancer. 2019 May 15. doi: 10.1002/cncr.32175. [Epub ahead of print]
Patterson WB. The quality of survival in response to treatment. JAMA. 1975;233(3):280-281. doi:10.1001/jama.1975.03260030070034
van Dam FS, Somers R, van Beek-Couzijn AL. Quality of life: some theoretical issues. J. Clin. Pharmacol. 1981. 21(Suppl. 8–9), 166S–168S (1981). doi.org/10.1002/j.1552-4604.1981.tb02592.x
Ira B. Wilson, MD, MSc; Paul D. Cleary, PhD. Linking Clinical Variables With Health-Related Quality of Life A Conceptual Model of Patient Outcomes JAMA. 1995;273(1):59-65. doi:10.1001/jama.1995.03520250075037
Kluetz PG, Papadopoulos EJ, Johnson LL, Donoghue M, Kwitkowski VE, Chen WH, Sridhara R, Farrell AT, Keegan P, Kim G, Pazdur R. Focusing on Core Patient-Reported Outcomes in Cancer Clinical Trials-Response. Clin Cancer Res. 2016 Nov 15;22(22):5618. doi: 10.1158/1078-0432.CCR-16-2140.
Zettler M, Basch E, Nabhan C. Surrogate End Points and Patient-Reported Outcomes for Novel Oncology Drugs Approved Between 2011 and 2017. JAMA Oncol. 2019 Jul 3. doi: 10.1001/jamaoncol.2019.1760. [Epub ahead of print]
Basch E, Reeve BB, Mitchell SA, Clauser SB, Minasian LM, Dueck AC, Mendoza TR, Hay J, Atkinson TM, Abernethy AP, Bruner DW, Cleeland CS, Sloan JA, Chilukuri R, Baumgartner P, Denicoff A, St Germain D, O’Mara AM, Chen A, Kelaghan J, Bennett AV, Sit L, Rogak L, Barz A, Paul DB, Schrag D. Development of the National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). J Natl Cancer Inst. 2014 Sep 29;106(9):dju244. doi: 10.1093/jnci/dju244. PubMed PMID: 25265940; PubMed Central PMCID: PMC4200059.
Image Credits
Modified from original https://health.mil/-/media/Images/MHS/Photos/LT-Col-Owen-Johnson-plastic-surgeon.ashx

