Sara Musetti
When someone says the word “cancer.” it can tie knots in the guts of even the strongest among us. There is no pretending that cancer isn’t a terrifying phenomenon that has touched most of our lives. Very few cancers, however, are able to elicit the chill that brain cancer does, in patients, family members, and researchers alike. Most often, tumors found in the brain originated in other organs, like the breast or lungs. But several types originate in the brain, varying by location and deadliness.
One of the biggest challenges in treating brain tumors is, of course, the location itself. Surgery remains the most effective way of removing cancerous tissue throughout the body, but the brain is so delicate that it can be difficult to remove the entirety of the tumor without doing significant damage to a patient’s function. Radiation can also be risky, although improvements in the way radiation beams are targeted continue to give patients better and better outcomes. However, for aggressive brain cancers like glioblastoma, which recently lead to the death of US Senator John McCain, the best many patients can hope for is a post-diagnosis survival measured in months. Recently, Kaye Alcedo wrote a comprehensive piece on why glioblastoma is such a tricky disease. Complicating matters further is the blood-brain barrier (BBB). Because the brain is so precious and delicate, the cells that form the interface between the bloodstream and brain tissue form a tight barrier that prohibits everything but the smallest molecules from crossing without the help of transporters and receptors. This keeps your brain safe from bacteria or toxins that might be in your blood, reducing infections that could shut down your body’s function from the command center. Immune cells, necessary for fighting infection and cancer, can occasionally cross with the help of specialized receptors, but that is tightly regulated. Many of our most recent cancer breakthroughs, like immunotherapy, rely on large molecules or cells to reach the tumor, and they simply cannot cross the blood-brain barrier. At least, they couldn’t until now.

Heba Samaha and colleagues out of Baylor Medical Center were concerned with the difficulty of using immunotherapies that use T cells to treat cancer, previously discussed here on OncoBites, to treat brain tumors. The movement of immune cells from the bloodstream into any site of disease, whether it be a tumor, wound, or infection, relies on their interaction with proteins on the surface of blood vessels called cell-adhesion molecules (CAMs). The same is true for the blood-brain barrier. Generally, T cells access the brain through two waves of interactions with different CAMs; first, they bind to activated leukocyte cell adhesion molecule (ALCAM), which slows them down before they bind to ICAM1 (intercellular adhesion molecule) or VCAM1 (vascular adhesion molecule 1), further aiding exit from the bloodstream. The Baylor team found that while ALCAM was still expressed in the tumor-adjacent blood-brain barrier (or blood-brain tumor barrier), ICAM and VCAM are almost nonexistent. This lack of “second-wave” cell adhesion molecules appears to be what keeps immune cells from reaching brain tumors, limiting the effectiveness of many immunotherapies. Therefore, the team from Baylor hypothesized that by using gene editing to alter T cells used in immunotherapy, they could enable those cells to cross the BBB through interactions with ALCAM alone and therefore avoid challenges associated with the lack of ICAM and VCAM near the tumor.
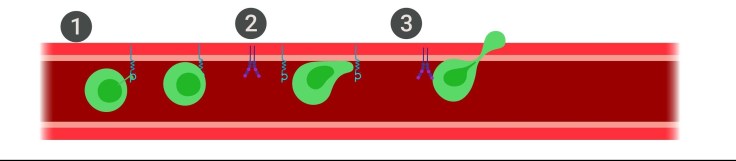
T cells, the cancer-cell killing immune cells, bind with ALCAM on the BBB through a protein on their surface called CD6. Samaha and her team created a form of CD6 that binds with ALCAM through more than one site, increasing the strength and endurance of the interaction. After editing the genome of T cells to express this rationally designed protein, they found that their engineered cells were able to cross the BBB in two different brain cancer models where ALCAMs existed alone, enabling them to reach the tumor in much greater numbers than seen using normal T cells. They also found that they could make these cells into CAR-T cells, which are capable of specifically targeting and killing the tumor types they used as their models.
As T cell engineering is not uncommon in cancer therapy, and was in fact recently approved by the FDA, improvements such as these to T cells to make them capable of crossing the blood-brain barrier is a significant step to making brain tumors more accessible to treatment. However, this technology is so far only successful in mice, and requires a great deal more work and study before making its way into human patients. It is a reminder, though, of the promise ahead of us in targeted cancer research, and of the ingenuity of researchers in the fact of seemingly insurmountable odds.
References
Samaha, H., Pignata, A., Fousek, K., Ren, J., Lam, F.W., Stossi, F., Dubrulle, J., Salsman, V.S., Krishnan, S., Hong, S.H. and Baker, M.L. (2018). A homing system targets therapeutic T cells to brain cancer. Nature.
Header image: Axial MRI scans of a patient with multiple sclerosis. © NIH Image Gallery.

