Yitong Li
How to treat cancers has always been one of the biggest challenges in modern medical practice. While current chemotherapies and radiotherapies have shown significant progress, not all patients survive treatment, and only a portion of those who survive do so without suffering from significant trauma. Hence, early diagnosis and safer treatments are the keys to our combat against the tumor.
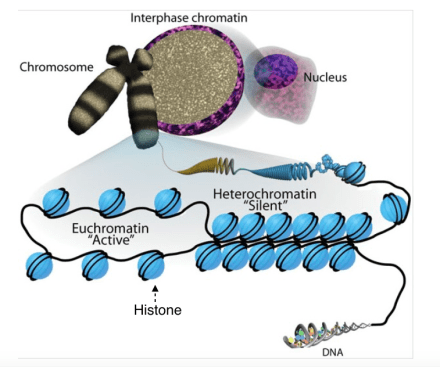
Working towards these goals, a research group in Australia have recently developed a new way to treat cancers without damaging cells. Dr. Tim Thomas and Dr. Anne Voss from the Walter and Eliza Hall Institute, the senior investigators who led the study, reported in Nature this past month that they have identified small molecule inhibitors that prevent tumor growth by inhibiting protein KAT6A and KAT6B. KAT6A and KAT6B are two enzymes that catalyze biochemical reactions involved in DNA regulation. By modifying histones, the key scaffolding proteins for DNA packing, KAT6A/B can regulate chromatin accessibility to transcription factors, which controls gene expression in the cell. Both KAT6A and KAT6B have been implicated in various tumor cases, such as acute myeloid leukemia, lymphoma, breast cancer, lung adenocarcinoma, and medulloblastoma. Therefore, as key pro-oncogenic factors, KAT6A/B appear to be good targets for cancer treatment.
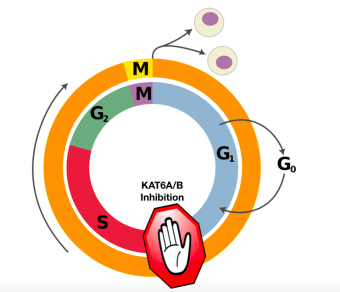
By selecting the synthetic molecules that specifically bind to KAT6A/B, the researchers identified multiple molecules that inhibit KAT6A/B activity. Some of these molecules are stable in the blood and harmless to animals, making them possible drug candidates for human patients. Given that KAT6A/B promotes cell division, applying these inhibitors to cells successfully prevented the cells from entering the cell cycle, the unstoppable journey that will only lead to the formation of two daughter cells once started. Most tumor cells have lost their checkpoints for entry into the cell cycle, thus left dividing indefinitely. Gating cell cycle entry can potentially halt tumor growth by limiting unregulated cell division in the tumor tissues. Testing these KAT6A/B inhibitors in animal models, the researchers have demonstrated the potency of one molecule in treating lymphoma in mice without any observable side effects.
The concept of halting cell division is not new. In fact, most healthy cells in the body maintain a dormant state, in which signals promoting cell division are suppressed so cell division does not occur. This dormant state is called senescence, deriving from Latin senex meaning old. In cell biology, senescence is usually used to refer to the cessation of cell division. Senescent cells may still be alive and functional for a limited amount of time, but they won’t generate more daughter cells. In the case of cancer, dysregulated cell division drives the formation of solid tumors. Hence, to fundamentally cure cancer, it is necessary to remove the over-duplicating cells and/or to prevent them from further
duplication. Multiple methods can be used to trigger cell death, and damaging DNA is one. Most chemo- and radio-therapies work by inducing structural breaks in the double-stranded DNA helices, which then initiate the suicidal programs in the cell. However, lacking specific targeting, chemo- and radio-therapies affects all rapidly dividing cells unbiasedly, killing both the tumor cells and the precious stem cells in our body. While most healthy cells don’t actively divide in our body, stem cells are required to constantly progress through the cell cycle to replace dead cells on our skin, scalp, the surface of our intestine, and in many more organs. As proper duplication of the stem cell populations is the key to many physiological activities, patients receiving these treatments tend to lose hair, develop various GI problems, and become susceptible to infections easily. Additionally, DNA-damaging agents can also lead to the accumulation of genetic mutations, which is capable of turning healthy cells into another tumor mass in the future. Therefore, this new discovery that can halt tumor growth without damaging the genetic materials smartly circumvents most of the side effects from current standard cancer treatments, lighting up the hope of recovering from tumors.
The first to target cell cycle progression without introducing DNA damage, these inhibitors present great potential as a new class of cancer treatments. By stalling tumor growth, inhibiting KAT6A/B can provide the doctor and patient extra time to figure out the ultimate treatment. For severely sick patients, every extra second count. Although these inhibitors still need to be thoroughly validated in clinical trials and an efficient drug-delivering system is required for targeting tumor cells, Dr.Thomas and Dr. Voss’ groups have demonstrated the feasibility and efficacy of targeting cell cycle regulation to prevent tumor growth. By bring reporting this new discovery, they have ignited the hope of finding a cure for cancers by putting the bad cells in deep sleep.
Work Discussed
Baell, J. B., Leaver, D. J., Hermans, S. J., Kelly, G. L., Brennan, M. S., Downer, N. L., . . . Thomas, T. (2018). Inhibitors of histone acetyltransferases KAT6A/B induce senescence and arrest tumour growth. Nature, 560(7717), 253-257. doi: 10.1038/s41586-018-0387-5
Image Credits
A schematic drawing of DNA packing in the cell. Image adapted from Wikipedia
A schematic drawing of KAT6A/B inhibition preventing cells from entering the cell cycle. Image adapted from Wikimedia


Leave a comment