Reading time: 5 minutes
Sandhyaa Venkatachalam
Treating cancers, which can be as unpredictable as loose cannons, is an uphill battle even now in the age of modern medicine. Most cancer treatments rely on chemotherapy, which uses powerful drugs to kill cancer cells. However, chemotherapy has significant limitations. These drugs not only target cancerous cells but can also damage nearby healthy tissues, leading to severe side effects such as nausea, heart damage, and immune suppression.
Scientists are investigating methods to protect healthy tissues by delivering chemotherapy drugs directly to tumors, thereby reducing tissue damage. One promising method involves nanomedicine, an application where very tiny tools (less than 100 – 1000 nanometers) are used to fix healthy issues. On that note, some labs are actively studying plant-based tiny delivery systems known as ‘nanodrug carriers.’ These delivery systems are safe, biodegradable, non-toxic, high yielding, and cost effective. They can transport drugs directly to a specific target in the body, similar to a delivery vehicle bringing a package to a specific address.
Lemon Juice-derived Vesicle Delivery System
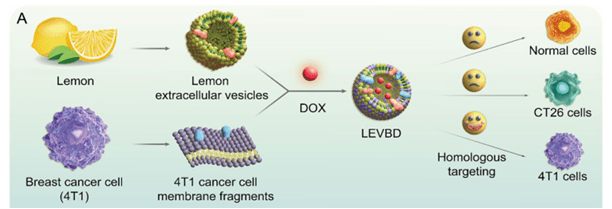
According to the Journal of Nanobiotechnology (2025), Yang LU et al. focused on tiny particles found naturally in lemon juice, known as lemon-derived extracellular vesicles or LEVs, as the base material for their drug carrier. They decided to work with lemon vesicles over other plant-based vesicles such as yams or grapefruit, which had been previously tested by earlier researchers and yet fell short on site-specific targeting and/or effectiveness. Lemons are also known for their anti-cancer potential.
Plants typically use these tiny, bubble-shaped vesicles/sacs to transfer signals and protein cargo between their cells. However, it is difficult for LEVs to identify tumor cells on their own because they lack the “address label” that guides them to the tumor’s exact location.
Therefore, as illustrated in Figure 1 below, scientists used outer membrane fragments from a type of breast cancer cell, 4T1, and fused them onto the surface of lemon vesicles using a sonicator that generates high-frequency sound waves. This was because cancer cell membranes have distinct adhesion proteins on their surfaces that allow them to identify and attach to other cancer cells of the same type – an incredible phenomenon called ‘homologous targeting.’ The researchers, therefore, hypothesized that combining LEVs with cancer cell membrane components would enable the vesicles to bind specifically to tumors of the same type.
The hybrid vesicles were loaded with doxorubicin (DOX), a commonly used anti-cancer drug. The final composite nanodrug delivery vector, called LEVBD (lemon-derived vesicle, breast cancer cell membrane, and doxorubicin), is designed to circulate in the bloodstream, target 4T1 tumors, attach to them, and then release the drug directly into the cancer tissues.
Assessing Functional Efficacy of Vesicles
To test whether the constructed “vehicle” actually functions, Yang LU’s team conducted several experiments. First, they used specialized imaging and protein analysis techniques to confirm if cancer cell membrane proteins had indeed merged with the lemon vesicles. Important surface protein markers such as CD44, CD47, and E-cadherin were found to be present. These are the strong adhesion proteins that help cancer cells to stick onto one another.
They also discovered that a high amount of doxorubicin could be packed into these vesicles, which meant that LEVBD had a high drug-loading efficiency. Interestingly, they found that these vesicles released the drug faster in acidic conditions. Tumors typically create a slightly more acidic environment than healthy tissues by producing large amounts of lactic acid, so as to hinder the function of immune cells. Therefore, this helps the vesicles in releasing the drug more efficiently at the site of the tumor.
Next, researchers examined vesicle reactions in living cells and animals.
When they placed LEVBD in dishes containing 4T1 and CT26 colon cancer cells, they found that 4T1 cells absorbed more vesicles than CT26 cells. (Figure 1)
Then the most convincing evidence came from experiments using mice, as shown in Figure 2. LEVBD were injected into mice with 4T1 breast tumors, and fluorescent markers were used to track the particles. They found that the hybrid vesicles accumulated mainly in tumors rather than in other organs such as the liver, kidney, or spleen. To further test specificity, they used mice that carried two different types of tumors: one with 4T1 breast cancer cells and the other with CT26 colon cancer cells. The vesicles were mainly concentrated in the 4T1 tumor cells, indicating homologous targeting.
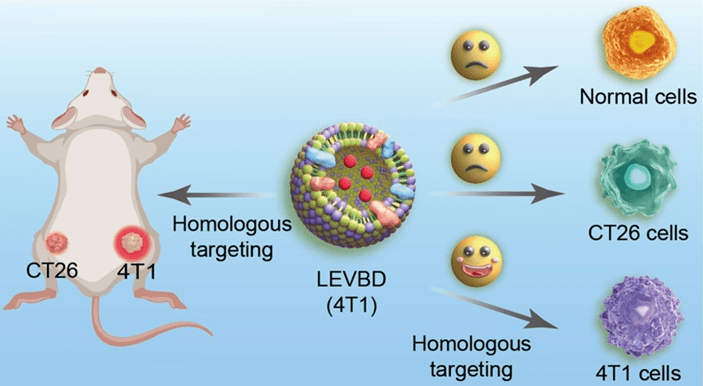
Mice treated with LEVBD showed a significant reduction in tumor growth compared to mice that received either free doxorubicin or unmodified lemon vesicles without the cancer membrane coating. Moreover, the treated mice showed no obvious signs of toxicity in other organs as checked under a microscope, and their blood test results (white blood cells, red blood cells, hemoglobin, platelets) reflected numbers within healthy ranges.
Discussion
Overall, while we have lemon vesicles that are biocompatible on the one hand, and cancer cell membranes, which contain specific proteins on the other – both when combined, can form a “smart” delivery system that helps them identify tumor cells in the body, bearing the same specific markers, as provided on their outer membranes.
Research on the use of LEVs for cancer treatment is still in its early stages, and many questions surrounding the use of LEVs on human cells must be investigated. Researchers must determine whether this phenomenon can be repeated for other cancer types and further develop practical solutions to scale up vesicle production for clinical use. Despite challenges, this method offers a new ray of hope for curing cancers and a roadmap for the future of nanomedicine.
Header Image source: www.pexels.com
Figure 1 source: Scheme 1 from Yang et al, Journal of Nanobiotechnology (2025) 23:74
Figure 2 source: Graphical abstract from Yang et al., Journal of Biotechnology (2025) 23:74
Edited by Joycelyn Ghansah
Reference:
Yang LY, Liang GW, et al. Enhanced tumor self-targeting of lemon-derived extracellular vesicles by embedding homotypic cancer cell membranes for efficient drug delivery. Journal of Nanobiotechnology (2025) 23:74.


Leave a comment